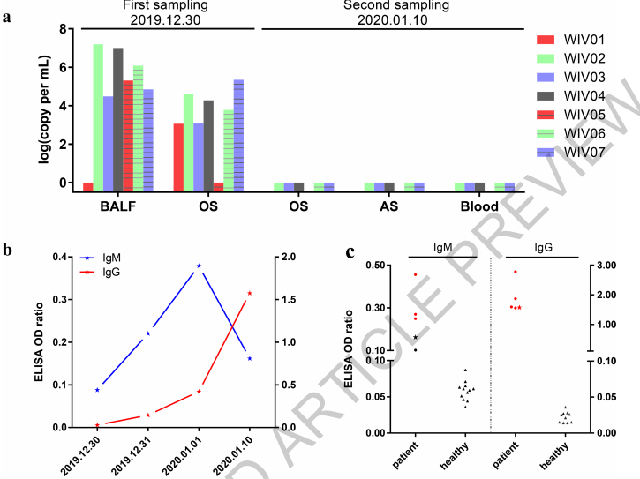
Post by Admin on Feb 7, 2020 22:50:09 GMT
A novel coronavirus (2019-nCov) was identified in Wuhan, Hubei Province,
China in December of 2019. This new coronavirus has resulted in thousands of
cases of lethal disease in China, with additional patients being identified in a
rapidly growing number internationally. 2019-nCov was reported to share the
same receptor, Angiotensin-converting enzyme 2 (ACE2), with SARS-Cov.
Here based on the public database and the state-of-the-art single-cell RNASeq
technique, we analyzed the ACE2 RNA expression profile in the normal
human lungs. The result indicates that the ACE2 virus receptor expression is
concentrated in a small population of type II alveolar cells (AT2). Surprisingly,
we found that this population of ACE2-expressing AT2 also highly expressed
many other genes that positively regulating viral reproduction and transmission.
A comparison between eight individual samples demonstrated that the Asian
male one has an extremely large number of ACE2-expressing cells in the lung.
This study provides a biological background for the epidemic investigation of
the 2019-nCov infection disease, and could be informative for future anti-ACE2
therapeutic strategy development.
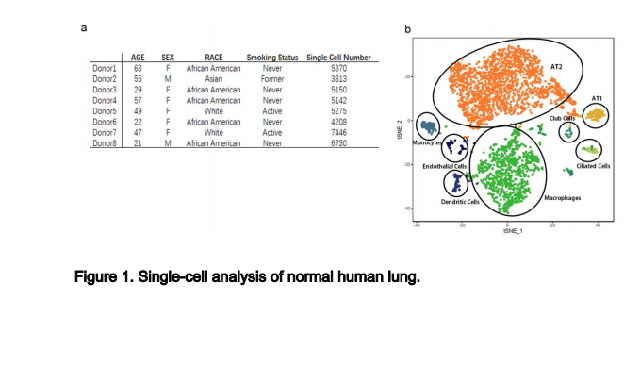
a. Characteristics of lung transplant donors for single-cell RNA-Seq analysis.
b. Cellular cluster map of the Asian male. All 8 samples were analyzed using
the Seurat R package. Cells were clustered using a graph-based shared
nearest neighbor clustering approach and visualized using a t-distributed
Stochastic Neighbor Embedding (tSNE) plot.
Severe infection by 2019-nCov could result in acute respiratory distress
syndrome (ARDS) and sepsis, causing death in approximately 15% of infected
individuals1,2. Once contacted with the human airway, the spike proteins of this
virus can associate with the surface receptors of sensitive cells, which mediated
the entrance of the virus into target cells for further replication. Recently, Xu
et.al., modeled the spike protein to identify the receptor for 2019-nCov, and
indicated that Angiotensin-converting enzyme 2 (ACE2) could be the receptor
for this virus3. ACE2 is previously known as the receptor for SARS-Cov and
NL634-6. According to their modeling, although the binding strength between
2019-nCov and ACE2 is weaker than that between SARS-Cov and ACE2, it is
still much higher than the threshold required for virus infection. Zhou et al.
conducted virus infectivity studies and showed that ACE2 is essential for 2019-
nCov to enter HeLa cells7. These data indicated that ACE2 is likely to be the
receptor for 2019-nCov The expression and distribution of the receptor decide
the route of virus nfection and the route of infection has a major implication for
understanding the pathogenesis and designing therapeutic strategies. Previous studies
have investigated the RNA expression of ACE2 in 72 human tissues8. However, the
lung is a complex organ with multiple types of cells, and such real-time PCR
RNA profiling is based on bulk tissue analysis with no way to elucidate the
ACE2 expression in each type of cell in the human lung. The ACE2 protein level
is also investigated by immunostaining in lung and other organs8,9. These
studies showed that in normal human lung, ACE2 is mainly expressed by type
II and type I alveolar epithelial cells. Endothelial cells were also reported to be
ACE2 positive. However, immunostaining analysis is known for its lack of signal
specificity, and accurate quantification is also another challenge for such
analysis.
The recently developed single-cell RNA sequencing (scRNA-Seq)
technology enables us to study the ACE2 expression in each cell type and give
quantitative information at single-cell resolution. Previous work has built up the
online database for scRNA-Seq analysis of 8 normal human lung transplant
donors10. In current work, we used the updated bioinformatics tools to analyze
the data. In total, we analyzed 43,134 cells derived from normal lung tissue of
8 adult donors. We performed unsupervised graph-based clustering (Seurat
version 2.3.4) and for each individual, we identified 8~11 transcriptionally
distinct cell clusters based on their marker gene expression profile. Typically
the clusters include type II alveolar cells (AT2), type I alveolar cells (AT1),
airway epithelial cells (ciliated cells and Club cells), fibroblasts, endothelial cells
and various types of immune cells. The cell cluster map of a representative
donor (Asian male, 55-year-old) was visualized using t-distributed stochastic
neighbor embedding (tSNE) as shown in Fig. 1b and his major cell type marker
expressions were demonstrated in Fig.2.
Next, we analyzed the cell-type-specific expression pattern of ACE2 in each
individual. For all donors, ACE2 is expressed in 0.64% of all human lung cells.
The majority of the ACE2-expressing cells (averagely 83%) are AT2 cells.
Averagely 1.4±0.4% of AT2 cells expressed ACE2. Other ACE2 expressing
cells include AT1 cells, airway epithelial cells, fibroblasts, endothelial cells, and
macrophages. However, their ACE2-expressing cell ratio is low and variable
among individuals. For the representative donor (Asian male, 55-year-old), the
expressions of ACE2 and cell-type-specific markers in each cluster are
demonstrated in Fig.2.
China in December of 2019. This new coronavirus has resulted in thousands of
cases of lethal disease in China, with additional patients being identified in a
rapidly growing number internationally. 2019-nCov was reported to share the
same receptor, Angiotensin-converting enzyme 2 (ACE2), with SARS-Cov.
Here based on the public database and the state-of-the-art single-cell RNASeq
technique, we analyzed the ACE2 RNA expression profile in the normal
human lungs. The result indicates that the ACE2 virus receptor expression is
concentrated in a small population of type II alveolar cells (AT2). Surprisingly,
we found that this population of ACE2-expressing AT2 also highly expressed
many other genes that positively regulating viral reproduction and transmission.
A comparison between eight individual samples demonstrated that the Asian
male one has an extremely large number of ACE2-expressing cells in the lung.
This study provides a biological background for the epidemic investigation of
the 2019-nCov infection disease, and could be informative for future anti-ACE2
therapeutic strategy development.

a. Characteristics of lung transplant donors for single-cell RNA-Seq analysis.
b. Cellular cluster map of the Asian male. All 8 samples were analyzed using
the Seurat R package. Cells were clustered using a graph-based shared
nearest neighbor clustering approach and visualized using a t-distributed
Stochastic Neighbor Embedding (tSNE) plot.
Severe infection by 2019-nCov could result in acute respiratory distress
syndrome (ARDS) and sepsis, causing death in approximately 15% of infected
individuals1,2. Once contacted with the human airway, the spike proteins of this
virus can associate with the surface receptors of sensitive cells, which mediated
the entrance of the virus into target cells for further replication. Recently, Xu
et.al., modeled the spike protein to identify the receptor for 2019-nCov, and
indicated that Angiotensin-converting enzyme 2 (ACE2) could be the receptor
for this virus3. ACE2 is previously known as the receptor for SARS-Cov and
NL634-6. According to their modeling, although the binding strength between
2019-nCov and ACE2 is weaker than that between SARS-Cov and ACE2, it is
still much higher than the threshold required for virus infection. Zhou et al.
conducted virus infectivity studies and showed that ACE2 is essential for 2019-
nCov to enter HeLa cells7. These data indicated that ACE2 is likely to be the
receptor for 2019-nCov The expression and distribution of the receptor decide
the route of virus nfection and the route of infection has a major implication for
understanding the pathogenesis and designing therapeutic strategies. Previous studies
have investigated the RNA expression of ACE2 in 72 human tissues8. However, the
lung is a complex organ with multiple types of cells, and such real-time PCR
RNA profiling is based on bulk tissue analysis with no way to elucidate the
ACE2 expression in each type of cell in the human lung. The ACE2 protein level
is also investigated by immunostaining in lung and other organs8,9. These
studies showed that in normal human lung, ACE2 is mainly expressed by type
II and type I alveolar epithelial cells. Endothelial cells were also reported to be
ACE2 positive. However, immunostaining analysis is known for its lack of signal
specificity, and accurate quantification is also another challenge for such
analysis.
The recently developed single-cell RNA sequencing (scRNA-Seq)
technology enables us to study the ACE2 expression in each cell type and give
quantitative information at single-cell resolution. Previous work has built up the
online database for scRNA-Seq analysis of 8 normal human lung transplant
donors10. In current work, we used the updated bioinformatics tools to analyze
the data. In total, we analyzed 43,134 cells derived from normal lung tissue of
8 adult donors. We performed unsupervised graph-based clustering (Seurat
version 2.3.4) and for each individual, we identified 8~11 transcriptionally
distinct cell clusters based on their marker gene expression profile. Typically
the clusters include type II alveolar cells (AT2), type I alveolar cells (AT1),
airway epithelial cells (ciliated cells and Club cells), fibroblasts, endothelial cells
and various types of immune cells. The cell cluster map of a representative
donor (Asian male, 55-year-old) was visualized using t-distributed stochastic
neighbor embedding (tSNE) as shown in Fig. 1b and his major cell type marker
expressions were demonstrated in Fig.2.
Next, we analyzed the cell-type-specific expression pattern of ACE2 in each
individual. For all donors, ACE2 is expressed in 0.64% of all human lung cells.
The majority of the ACE2-expressing cells (averagely 83%) are AT2 cells.
Averagely 1.4±0.4% of AT2 cells expressed ACE2. Other ACE2 expressing
cells include AT1 cells, airway epithelial cells, fibroblasts, endothelial cells, and
macrophages. However, their ACE2-expressing cell ratio is low and variable
among individuals. For the representative donor (Asian male, 55-year-old), the
expressions of ACE2 and cell-type-specific markers in each cluster are
demonstrated in Fig.2.