Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding
Nature Medicine volume 26, 502–505(2020)
Main
The outbreak of SARS-CoV-2 (formerly 2019-nCoV) infection emerged in December 2019 in Wuhan, Hubei Province, China1,2. By 25 February 2020, there had been 77,780 confirmed cases including 2,666 deaths in China and over 2,459 confirmed cases in 33 other countries3.
The genome of the new virus and early epidemiological and clinical features of the infection in adults have been reported4,5,6. The infection is estimated to have a mean incubation period of 5.2 d and commonly causes fever, cough, myalgia and pneumonia in patients4. To date there is a paucity of information regarding SARS-CoV-2 infection in children.
We here report the epidemiological and clinical features of ten children infected with SARS-CoV-2 and tested for evidence of viral excretion through the gastrointestinal and respiratory tracts.
By 20 February 2020, a total of 745 children and 3,174 adults, most of whom had either close contact with diagnosed patients or had members of the family reporting familial outbreaks in the previous 2 weeks, were screened by nasopharyngeal swab real-time PCR with reverse transcription (RT–PCR) for SARS-CoV-2 infection. Overall, 10 children (1.3%) and 111 adults (3.5%) tested positive. The 2.7-fold difference between children and adults is statistically significant (P = 0.002). All ten pediatric patients were admitted to our hospital, a treatment center for SARS-CoV-2 infection designated by the local municipal government.
Of the ten patients, six were male and four were female, with ages ranging from 2 months to 15 years (Table 1). Four had definite contact history with a confirmed patient, seven were from families with a cluster of infection and seven had travel history to epidemic areas in Hubei Province 2 weeks before the onset of infection.
Upon admission, seven had fever but none had a temperature above 39 °C (Table 1). Other symptom presentations included coughing (five children), sore throat (four children), nasal congestion and rhinorrhea (two children) and diarrhea (three children). One child was completely asymptomatic (patient 4). None of the patients had other symptoms commonly seen in adult patients such as lethargy, dyspnea, muscle ache, headache, nausea and vomiting and disorientation. In fact, none of them sought medical care; they were all identified and diagnosed because of their exposure history.
Chest X-rays of these patients were either normal or showed only coarse lung markings without unilateral or bilateral pneumonia. Chest computed tomography (CT) scans showed isolated or multiple patchy ground-glass opacities in five patients but were within normal ranges in the other five. These changes were mainly seen in the outer lung fields and few patients had subpleural bands or strips. There was no pleural effusion, enlarged lymph nodes or other changes that are typically seen in the critically ill adult patients4,6 (Supplementary Fig. 1).
Complete blood count, urine and stool analyses, coagulation function, blood biochemistry and infection biomarkers were tested upon admission (Table 1). Almost all test results were normal in the patients except for patient 9. Few cases had leukopenia, leukocytosis, lymphopenia or elevated transaminase, which in contrast are frequently seen in adult patients. Viral testing for influenza-A virus (H1N1, H3N2, H7N9), influenza B virus, respiratory syncytial virus, parainfluenza virus, adenovirus, SARS-CoV and MERS-CoV were negative in all patients.
Out of nine patients (the tenth patient was transferred from another hospital that did not conduct cytokine tests upon admission), seven showed elevated interleukin (IL)-17F and five of these patients had concurrent elevation of IL-22 (Supplementary Table 1). Five patients had elevated IL-6.
All patients received antiviral therapy with α-interferon oral spray initiated from admission (8,000 U, two sprays, three times a day). Patient 1 was the very first pediatric case of SARS-CoV-2 infection diagnosed in Guangzhou and also treated with azithromycin 10 mg kg−1 per day for 5 d and IVIG 300 mg kg−1 per day for 3 d. No patient required respiratory support or intensive care unit care.
We followed the pattern of viral excretion from respiratory and gastrointestinal tracts in all ten patients by a chronological series of nasopharyngeal and rectal swab samples using real-time RT–PCR (Fig. 1a). Patient 4 was asymptomatic but tested positive on multiple occasions. Patient 6 was asymptomatic on the day his nasopharyngeal swab tested positive and then developed nasal congestion and rhinorrhea the following day. The remaining eight patients had positive tests soon after the onset of symptoms. In addition, eight of ten patients also had real-time RT–PCR-positive rectal swabs, suggesting potential fecal viral excretion. Moreover, eight of ten patients (patients 1–6, 8 and 10) demonstrated persistently positive real-time RT–PCR tests of rectal swabs after their nasopharyngeal testing had become negative.
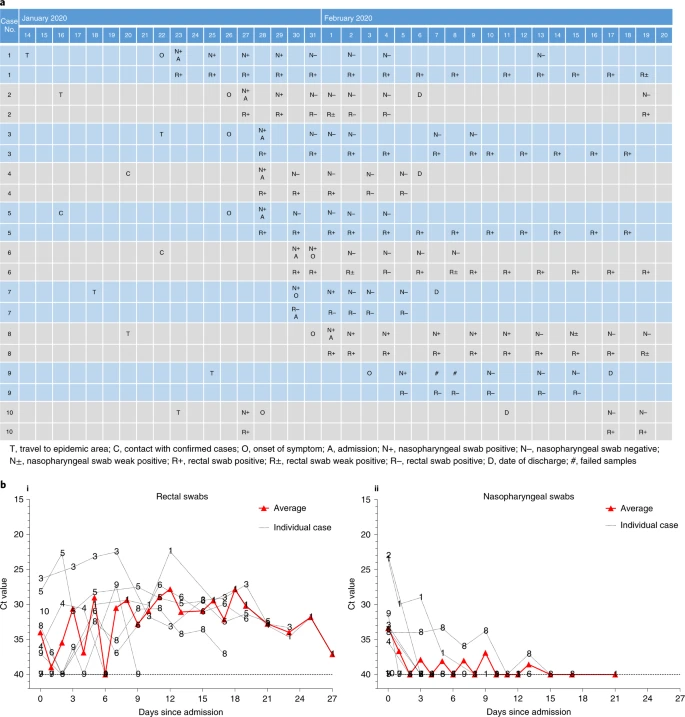
Fig. 1: Chronology of major epidemiological events and molecular testing results of n = 10 independent pediatric patients confirmed with SARS-CoV-2 infection.
a, Dates of exposure, illness onset and sampling and real-time RT–PCR results of nasopharyngeal swabs and rectal swabs. The total number of patients was n = 10 and real-time RT–PCR was assayed only once for one type of sample at one time point from one independent patient. Colors in the figure represent individual patients. b, Chronological changes in Ct values of Orf1ab and N genes using real-time RT–PCR after hospital admission. The Ct values of Orf1ab and N genes on real-time RT–PCR detected in rectal swabs obtained from n = 10 independent patients (i) and Ct values in nasopharyngeal swabs from n = 10 independent cases (ii). The Ct value is supposed to be inversely related to viral RNA copy numbers and a value of 40 means the virus is molecularly undetectable.
Patients 2, 4, 7 and 10 were discharged home after two consecutive negative real-time RT–PCR tests (separated by at least 24 h) on rectal swabs. Their nasopharyngeal and rectal swabs were repeated weekly after discharge. Patients 4 and 7 remained negative during follow-up but patient 2 had a positive rectal swab again 13 d after discharge. Interestingly, the mother of patient 2, who was hospitalized for SARS-CoV-2 infection (COVID-19) and discharged from a different hospital during the same time period, also had a positive rectal swab test on the same day as her child. A similar phenomenon was also observed in patient 10. He was hospitalized in the Eighth People’s Hospital of Guangzhou between 27 January and 11 February and discharged after two consecutive negative results for both nasopharyngeal and rectal swabs obtained 24 h apart. He was however re-admitted to our hospital because his rectal swab test became positive again on 17 February, although his nasopharyngeal swab test remained negative. As of 20 Feb, all remaining patients (patients 1, 3, 5, 6, 8 and 9) were still testing positive for rectal swabs and continued to be under hospital isolation and observation, although they had all recovered from their illness and had become asymptomatic.
As suggested in a recent study on a similar topic7, we used the cycle threshold (Ct) values of the serial rectal and nasopharyngeal swab tests to approximately indicate viral load (inversely related to Ct value) in these patients to show its change over time. Viral RNA measurements suggest that viral shedding from the digestive system might be greater and last longer than that from the respiratory tract (Fig. 1b).
Clinical features of SARS-CoV-2 infection in adults have been reported elswhere4,8,9. However, few pediatric cases are published and their clinical features have yet to be documented. Compared to adult patients, the ten pediatric patients reported here had clinically milder symptoms and showed fewer alterations in radiological and laboratory testing parameters. For example, none of the ten patients showed clear clinical signs or chest X-ray findings consistent with pneumonia, a typical feature seen in the initial adult patients4,6. Mild and atypical presentations of the infection in children may make it difficult to detect. Indeed, all the patients reported here were found through screening of suspected cases.
We also observed positive real-time RT–PCR results in rectal swabs in eight out of ten pediatric patients, which remained detectable well after nasopharyngeal swabs turned negative, suggesting that the gastrointestinal tract may shed virus and fecal–oral transmission may be possible. Indeed, fecal–oral transmission does exist with other respiratory viruses10. These findings also suggest that rectal swab-testing may be more useful than nasopharyngeal swab-testing in judging the effectiveness of treatment and determining the timing of termination of quarantine12. However, we do not have evidence of replication-competent virus in fecal swabs, which is required to confirm the potential for fecal–oral transmission.