|
|
Post by Admin on Nov 27, 2020 6:13:19 GMT
Results Participants A total of 17 258 Danish citizens responded to recruitment, and 6024 completed the baseline survey and fulfilled eligibility criteria. The first participants (group 1; n = 2995) were randomly assigned on 12 April 2020 and were followed from 14 to 16 April through 15 May 2020. Remaining participants (group 2; n = 3029) were randomly assigned on 24 April 2020 and were followed from 2 to 4 May through 2 June 2020. A total of 3030 participants were randomly assigned to the recommendation to wear face masks, and 2994 were assigned not to wear face masks (Figure); 4862 participants (80.7%) completed the study. Table 1 shows baseline characteristics, which were well balanced between groups. Participants reported having spent a median of 4.5 hours per day outside the home. 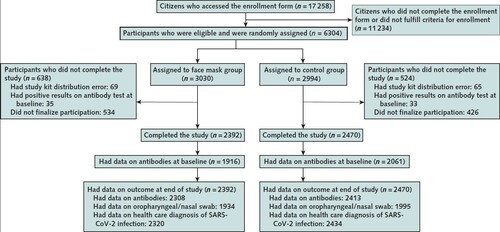 Inclusion and exclusion criteria are described in the Methods section, and criteria for completion of the study are given in the Supplement. SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2. 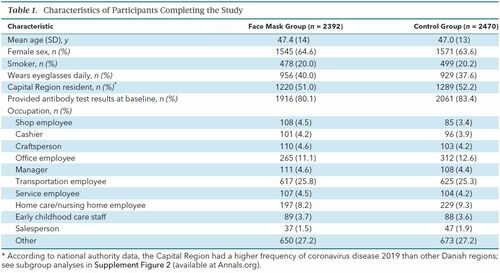 Table 1. Characteristics of Participants Completing the Study Adherence Based on the lowest adherence reported in the mask group during follow-up, 46% of participants wore the mask as recommended, 47% predominantly as recommended, and 7% not as recommended. |
|
|
|
Post by Admin on Nov 27, 2020 20:47:25 GMT
Primary Outcome The primary outcome occurred in 42 participants (1.8%) in the mask group and 53 (2.1%) in the control group. In an intention-to-treat analysis, the between-group difference was −0.3 percentage point (CI, −1.2 to 0.4 percentage point; P = 0.38) (odds ratio [OR], 0.82 [CI, 0.54 to 1.23]; P = 0.33) in favor of the mask group (Supplement Figure 1). When this analysis was repeated with multiple imputation for missing data due to loss to follow-up, it yielded similar results (OR, 0.81 [CI, 0.53 to 1.23]; P = 0.32). Table 2 provides data on the components of the primary end point, which were similar between groups. 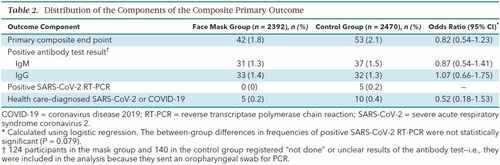 Table 2. Distribution of the Components of the Composite Primary Outcome In a per protocol analysis that excluded participants in the mask group who reported nonadherence (7%), SARS-CoV-2 infection occurred in 40 participants (1.8%) in the mask group and 53 (2.1%) in the control group (between-group difference, −0.4 percentage point [CI, −1.2 to 0.5 percentage point]; P = 0.40) (OR, 0.84 [CI, 0.55 to 1.26]; P = 0.40). Supplement Figure 2 provides results of the prespecified subgroup analyses of the primary composite end point. No statistically significant interactions were identified. In the preplanned sensitivity analysis, those who had a positive result on an antibody test at 1 month but had not provided antibody results at baseline were considered to have had positive results at baseline (n = 18)—that is, they were excluded from the analysis. In this analysis, the primary outcome occurred in 33 participants (1.4%) in the face mask group and 44 (1.8%) in the control group (between-group difference, −0.4 percentage point [CI, −1.1 to 0.4 percentage point]; P = 0.22) (OR, 0.77 [CI, 0.49 to 1.22]; P = 0.26). Three post hoc (not preplanned) analyses were done. In the first, which included only participants reporting wearing face masks “exactly as instructed,” infection (the primary outcome) occurred in 22 participants (2.0%) in the face mask group and 53 (2.1%) in the control group (between-group difference, −0.2 percentage point [CI, −1.3 to 0.9 percentage point]; P = 0.82) (OR, 0.93 [CI, 0.56 to 1.54]; P = 0.78). The second post hoc analysis excluded participants who did not provide antibody test results at baseline; infection occurred in 33 participants (1.7%) in the face mask group and 44 (2.1%) in the control group (between-group difference, −0.4 percentage point [CI, −1.4 to 0.4 percentage point]; P = 0.33) (OR, 0.80 [CI, 0.51 to 1.27]; P = 0.35). In the third post hoc analysis, which investigated constellations of patient characteristics, we did not find a subgroup where face masks were effective at conventional levels of statistical significance (data not shown). A total of 52 participants in the mask group and 39 control participants reported COVID-19 in their household. Of these, 2 participants in the face mask group and 1 in the control group developed SARS-CoV-2 infection, suggesting that the source of most observed infections was outside the home. Reported symptoms did not differ between groups during the study period (Supplement Table 3). Secondary Outcomes In the mask group, 9 participants (0.5%) were positive for 1 or more of the 11 respiratory viruses other than SARS-CoV-2, compared with 11 participants (0.6%) in the control group (between-group difference, −0.1 percentage point [CI, −0.6 to 0.4 percentage point]; P = 0.87) (OR, 0.84 [CI, 0.35 to 2.04]; P = 0.71). Positivity for any virus, including SARS-CoV-2, occurred in 9 mask participants (0.5%) versus 16 control participants (0.8%) (between-group difference, −0.3 percentage point [CI, −0.9 to 0.2 percentage point]; P = 0.26) (OR, 0.58 [CI, 0.25 to 1.31]; P = 0.19). |
|
|
|
Post by Admin on Nov 27, 2020 22:27:36 GMT
Discussion
In this community-based, randomized controlled trial conducted in a setting where mask wearing was uncommon and was not among other recommended public health measures related to COVID-19, a recommendation to wear a surgical mask when outside the home among others did not reduce, at conventional levels of statistical significance, incident SARS-CoV-2 infection compared with no mask recommendation. We designed the study to detect a reduction in infection rate from 2% to 1%. Although no statistically significant difference in SARS-CoV-2 incidence was observed, the 95% CIs are compatible with a possible 46% reduction to 23% increase in infection among mask wearers. These findings do offer evidence about the degree of protection mask wearers can anticipate in a setting where others are not wearing masks and where other public health measures, including social distancing, are in effect. The findings, however, should not be used to conclude that a recommendation for everyone to wear masks in the community would not be effective in reducing SARS-CoV-2 infections, because the trial did not test the role of masks in source control of SARS-CoV-2 infection. During the study period, authorities did not recommend face mask use outside hospital settings and mask use was rare in community settings (22). This means that study participants' exposure was overwhelmingly to persons not wearing masks.
The observed infection rate was similar to that reported in other large Danish studies during the study period (26, 30). Of note, the observed incidence of SARS-CoV-2 infection was higher than we had estimated when planning a sample size that would ensure more than 80% power to detect a 50% decrease in infection. The intervention lasted only 1 month and was carried out during a period when Danish authorities recommended quarantine of diagnosed patients, physical distancing, and hand hygiene as general protective means against SARS-CoV-2 transmission (23). Cafés and restaurants were closed through 18 May, but follow-up of the second randomized group continued through 2 June.
The first randomized group was followed while the Danish society was under lockdown. Reopening occurred (18 May 2020) during follow-up of the second group of participants, but it was not reflected in the outcome because infection rates were similar between groups (Supplement Figure 2). The relative infection rate between mask wearers and those not wearing masks would most likely be affected by changes in applied protective means or in the virulence of SARS-CoV-2, whereas the rate difference between the 2 groups would probably not be affected solely by a higher—or lower—number of infected citizens.
Although we saw no statistically significant difference in presence of other respiratory viruses, the study was not sufficiently powered to draw definite conclusions about the protective effect of masks for other viral infections. Likewise, the study had limited power for any of the subgroup analyses.
The primary outcome was mainly defined by antibodies against SARS-CoV-2. This definition was chosen because the viral load of infected patients may be only transiently detectable (31, 32) and because approximately half of persons infected with SARS-CoV-2 are asymptomatic (33, 26). Masks have been hypothesized to reduce inoculum size (34) and could increase the likelihood that infected mask users are asymptomatic, but this hypothesis has been challenged (35). For these reasons, we did not rely solely on identification of SARS-CoV-2 in oropharyngeal/nasal swab samples. As mentioned in the Methods section, an internal validation study estimated that the point-of-care test has 82.5% sensitivity and 99.5% specificity (26).
The observed rate of incident SARS-CoV-2 infection was similar to what was estimated during trial design. These rates were based on thorough screening of all participants using antibody measurements combined with PCR, whereas the observed official infection rates relied solely on PCR test–based estimates during the period. In addition, authorities tested only a small subset of primarily symptomatic citizens of the entire population, yielding low incidence rates. On this basis, the infection rates we report here are not comparable with the official SARS-CoV-2 infection rates in the Danish population. The eligibility requirement of at least 3 hours of exposure to other persons outside the home would add to this difference. Between 6 April and 9 May 2020, we found a similar seroprevalence of SARS-CoV-2 of 1.9% (CI, 0.8% to 2.3%) in Danish blood donors using the Livzon point-of-care test and assessed by laboratory technicians (36). Testing at the end of follow-up, however, may not have captured any infections contracted during the last part of the study period, but this would have been true in both the mask and control groups and was not expected to influence the overall findings.
The face masks provided to participants were high-quality surgical masks with a filtration rate of 98% (37). A published meta-analysis found no statistically significant difference in preventing influenza in health care workers between respirators (N95 [American standard] or FFP2 [European standard]) and surgical face masks (38). Adherence to mask use may be higher than observed in this study in settings where mask use is common. Some mask group participants (14%) reported adverse reactions from other citizens (Supplement Table 4). Although adherence may influence the protective effect of masks, sensitivity analyses had similar results across reported adherence.
How SARS-CoV-2 is transmitted—via respiratory droplets, aerosols, or (to a lesser extent) fomites—is not firmly established. Droplets are larger and rapidly fall to the ground, whereas aerosols are smaller (≤5 μm) and may evaporate and remain in the air for hours (39). Transmission of SARS-CoV-2 may take place through multiple routes. It has been argued that for the primary route of SARS-CoV-2 spread—that is, via droplets—face masks would be considered effective, whereas masks would not be effective against spread via aerosols, which might penetrate or circumnavigate a face mask (37, 39). Thus, spread of SARS-CoV-2 via aerosols would at least partially explain the present findings. Lack of eye protection may also have been of importance, and use of face shields also covering the eyes (rather than face masks only) has been advocated to halt the conjunctival route of transmission (40, 41). We observed no statistically significant interaction between wearers and nonwearers of eyeglasses (Supplement Figure 2). Recent reports indicate that transmission of SARS-CoV-2 via fomites is unusual (42), but masks may alter behavior and potentially affect fomite transmission.
The present findings are compatible with the findings of a review of randomized controlled trials of the efficacy of face masks for prevention (as personal protective equipment) against influenza virus (18). A recent meta-analysis that suggested a protective effect of face masks in the non–health care setting was based on 3 observational studies that included a total of 725 participants and focused on transmission of SARS-CoV-1 rather than SARS-CoV-2 (12). Of 725 participants, 138 (19%) were infected, so the transmission rate seems to be higher than for SARS-CoV-2. Further, these studies focused on prevention of infection in healthy mask wearers from patients with a known, diagnosed infection rather than prevention of transmission from persons in their surroundings in general. In addition, identified comparators (control participants) not wearing masks may also have missed other protective means. Recent observational studies that indicate a protective association between mandated mask use in the community and SARS-CoV-2 transmission are limited by study design and simultaneous introduction of other public health interventions (14, 43).
Several challenges regarding wearing disposable face masks in the community exist. These include practical aspects, such as potential incorrect wearing, reduced adherence, reduced durability of the mask depending on type of mask and occupation, and weather. Such circumstances may necessitate the use of multiple face masks during the day. In our study, participants used a mean of 1.7 masks per weekday and 1.3 per weekend day (Supplement Table 4). Wearing a face mask may be physically unpleasant, and psychological barriers and other side effects have been described (44). “Face mask policing” between citizens might reinforce use of masks but may be challenging. In addition, the wearer of a face mask may change to a less cautious behavior because of a false sense of security, as pointed out by WHO (17); accordingly, our face mask group seemed less worried (Supplement Table 4), which may explain their increased willingness to wear face masks in the future (Supplement Table 5). These challenges, including costs and availability, may reduce the efficacy of face masks to prevent SARS-CoV-2 infection.
The potential benefits of a community-wide recommendation to wear masks include combined prevention and source control for symptomatic and asymptomatic persons, improved attention, and reduced potential stigmatization of persons wearing masks to prevent infection of others (17). Although masks may also have served as source control in SARS-CoV-2–infected participants, the study was not designed to determine the effectiveness of source control.
The most important limitation is that the findings are inconclusive, with CIs compatible with a 46% decrease to a 23% increase in infection. Other limitations include the following. Participants may have been more cautious and focused on hygiene than the general population; however, the observed infection rate was similar to findings of other studies in Denmark (26, 30). Loss to follow-up was 19%, but results of multiple imputation accounting for missing data were similar to the main results. In addition, we relied on patient-reported findings on home antibody tests, and blinding to the intervention was not possible. Finally, a randomized controlled trial provides high-level evidence for treatment effects but can be prone to reduced external validity.
Our results suggest that the recommendation to wear a surgical mask when outside the home among others did not reduce, at conventional levels of statistical significance, the incidence of SARS-CoV-2 infection in mask wearers in a setting where social distancing and other public health measures were in effect, mask recommendations were not among those measures, and community use of masks was uncommon. Yet, the findings were inconclusive and cannot definitively exclude a 46% reduction to a 23% increase in infection of mask wearers in such a setting. It is important to emphasize that this trial did not address the effects of masks as source control or as protection in settings where social distancing and other public health measures are not in effect.
Reduction in release of virus from infected persons into the environment may be the mechanism for mitigation of transmission in communities where mask use is common or mandated, as noted in observational studies. Thus, these findings do not provide data on the effectiveness of widespread mask wearing in the community in reducing SARS-CoV-2 infections. They do, however, offer evidence about the degree of protection mask wearers can anticipate in a setting where others are not wearing masks and where other public health measures, including social distancing, are in effect. The findings also suggest that persons should not abandon other COVID-19 safety measures regardless of the use of masks. While we await additional data to inform mask recommendations, communities must balance the seriousness of COVID-19, uncertainty about the degree of source control and protective effect, and the absence of data suggesting serious adverse effects of masks (45).
This article was published at Annals.org on 18 November 2020
|
|
|
|
Post by Admin on Nov 28, 2020 20:39:45 GMT
The perspective of fluid flow behavior of respiratory droplets and aerosols through the facemasks in context of SARS-CoV-2 featured Physics of Fluids 32, 111301 (2020); doi.org/10.1063/5.0029767ABSTRACT In the unfortunate event of the current ongoing pandemic COVID-19, where vaccination development is still in the trial phase, several preventive control measures such as social distancing, hand-hygiene, and personal protective equipment have been recommended by health professionals and organizations. Among them, the safe wearing of facemasks has played a vital role in reducing the likelihood and severity of infectious respiratory disease transmission. The reported research in facemasks has covered many of their material types, fabrication techniques, mechanism characterization, and application aspects. However, in more recent times, the focus has shifted toward the theoretical investigations of fluid flow mechanisms involved in the virus-laden particles’ prevention by using facemasks. This exciting research domain aims to address the complex fluid transport that led to designing a facemask with a better performance. This Review discusses the recent updates on fluid flow dynamics through the facemasks. Key design aspects such as thermal comfort and flow resistance are discussed. Furthermore, the recent progress in the investigations on the efficacy of facemasks for the prevention of COVID-19 spread and the impact of wearing facemasks is presented. I. INTRODUCTION The person-to-person transmission of infectious respiratory diseases occurs primarily due to the transportation of virus-laden fluid particles from the infected person. The contagious fluid particles originate from the respiratory tract of the person and are expelled from the nose and the mouth during breathing, talking, singing, sneezing, and coughing.1–3 These particles have been broadly classified into two types: aerosols (aerodynamic particle size <5 μm) and droplets (aerodynamic particle size ≥5 μm–10 μm).4–6 The finding indicated that the transmission phenomena of these virus particles expelled by patients would be dependent on droplet sizes. Once expelled from the mouth or nose, larger respiratory droplets undergo gravitational settling before evaporation; in contrast, the smaller droplet particles evaporate faster than they settle, subsequently forming the aerosolized droplet nuclei that can be suspended for prolonged periods and travel in the air over long distances. The research studies have revealed that the severe acute respiratory syndrome (SARS) epidemic in 2003 and the current global pandemic of coronavirus disease 2019 (COVID-19) transmitted by contact or through the airborne route.7–10 Several preventive strategies such as safe distancing, contact tracing, isolation of the infected person, hand hygiene, and facemasks have been widely employed against the rapid spread of these diseases.11–14 Among them, the use of facemasks has proven to be one of the most effective protective measures against airborne virus transmission.15–20 The research suggested that face coverings could essentially reduce the forward distance traveled by a virus-laden droplet and thus has great potential to provide personal protection against airborne infection.21,22 Recently, the World Health Organization (WHO) has recommended using facemasks for the initial control of COVID-19 spread.23 In general, facemasks fall in the category of respiratory protection equipment (RPE) whose primary function is to protect the wearer from airborne viruses and contaminated fluids. There are various RPE types, ranging from simple homemade reusable cloth-based masks to surgical facemasks and N95 respirators to self-contained breathing apparatus.18,24–27 Different types of masks provide different levels of protection to the wearer. Surgical facemasks are loose-fitting, fluid-resistant, single-time use, and disposable, designed to cover the mouth and nose. These masks are fluid resistant and intended for reducing the emission of large respiratory droplets released during coughing and sneezing.28,29 However, there is a possibility of leakage around the facemask’s edge during the inhaling and exhaling processes. Such a dynamic leakage allows the direct contact of fluid droplets from the outside air to the wearer and vice versa. Such respiratory masks may also not provide adequate protection against extremely fine aerosolized particles, droplets, and nuclei.30 For efficient trapping of droplets, the facemask filters should contain microscopic pores; however, the minute-sized pores prevent air ventilation, which creates an uncomfortable situation for the wearer. Hence, a better trade-off between the pore sizes and the breathability is desirable for suitable facemasks. Some mask types that come with inbuilt respirators, such as a filtering facepiece respirator (FFR), P100 respirator/gas mask, self-contained breathing apparatus, full face respirator, and KN95 respirators, provide better breathability for the users. The name designation “N95” in the N95 respirators refers to the filtration of 0.3 µm sized particles with 95% efficiency.31 The filtration mechanism of N95 facemasks operates on three possible principles: diffusion, inertial impaction, and electrostatic attraction. The smaller particles (<1 µm) usually get diffused and stuck on the filter’s fibrous layers, whereas particles of typically ≥1 µm get influenced by the inertia effect, preventing them from flowing across the fibers in the filtration layers and get filtered. N95 masks are designed for single-use because of potential contamination of filter layers, resulting in rapid degradation of their filtration efficiency (FE). However, several innovative techniques have been demonstrated for decontaminating and reusing N95 masks.32,33 Some polymers such as polypropylene, polyethylene, polyesters, polyamides, polycarbonates, and polyphenylene oxide are usually utilized for the fabrication of N95 filter layers.34 However, some recent N95 respirator masks are fabricated using ionic surfactant coated electrocharged polymers or electrospun nanofibers as intermediate layers.35–37 These electret fibers trap particles through electrostatic or electrophoretic effects, which help in better filtration of small-size particle transmission.38–40 Because of the ongoing COVID-19 pandemic, a significant demand for facemasks has been reported worldwide while stimulating research about their efficacy for filtering expelled droplets from the infected person’s mouth and nose. In this regard, considerable efforts have been made in the past for the evaluation of facemasks’ performance. The quantitative performance of the facemasks has been typically characterized by evaluating the filtration efficiency (FE) and the total inward leakage (TIL).41–43 The filtration efficiency refers to the percentage of blocked particles by the tightly fitted facemasks. The filtration efficiency can be calculated as FE = (1 − (Cd/Cu)) × 100%, where Cu, Cd are the particle count in the upstream feed prior to filtration and in the downstream filtrate, respectively. TIL is defined as the percentage of particles entering the mask through both the filter and the leakage between the mask and the face. The total inward leakage is calculated by dividing the particle concentrations on the outside and inside the facemasks. The protection factor of the facemasks can be determined from the following expression: PF = 1/TIL. The higher PF value of the masks performs better in virus transmission control.16 Furthermore, the fluid penetration resistance performance of the facemasks has been evaluated as per the ASTM F1862/F1862M-17 standards.44,45 However, this test method does not evaluate facemasks’ performance for airborne exposure pathways or in preventing the penetration of aerosolized fluids deposited on the facemask. In recent times, some qualitative analysis has been demonstrated for the rapid design characterization of facemasks.46 While these experimental studies are essential for the broad characterization and design evaluation of respiratory facemasks, further theoretical and numerical methods and algorithm-based investigations provide a better insight into the facemask’s fluid flow dynamics and the droplet leakage through the facemask openings. If the facemask is donned for a prolonged period, the filtration efficiency may decrease due to the saturation effects.21 It has been usually neglected in experimental studies. To involve these factors, an alternative approach, the computational fluid dynamics (CFD) method, can be invaluable for understanding the fluid–particle flow behavior through the facemasks. The fluid dynamics-based numerical techniques have gained momentum in the field of the facemask research domain. The computational fluid flow models have shown their potentials in an improved prediction of the spreading of respiratory virus-laden droplets and aerosols, sensitive to the ambient environment and crucial to the public health responses.34 This Review focuses on the fluid flow aspects of the facemasks and their efficacy in virus transmission control. Following a brief introduction to the respiratory infectious diseases and their control strategies (Sec. I), the respiratory droplet transportation mechanisms in conjunction with the possible governing equations required for estimating the transport phenomena are presented in Sec. II. Then, the droplet transport behavior through the facemasks is described in Sec. III. Key design aspects for the facemasks are explained in Sec. IV. Section V covered the recent progress in investigating the efficacy of facemasks for preventing the virus spread. The impact of using the facemasks is discussed in Sec. VI. The concluding remarks and a brief outlook for future research directions are summarized in Sec. VII. |
|
|
|
Post by Admin on Nov 29, 2020 19:41:12 GMT
IV. KEY DESIGN ASPECTS A. Thermal comfort Thermal comfort is an essential aspect of a facemask as it may affect the compliance of the use of the facemask during summer or in tropical countries. There were reported incidence of skin rashes, increased heat stress,  , and discomfort due to prolonged wearing of a facemask in hot and humid conditions.70 To improve the thermal comfort level of facemasks, researchers have developed some unique facemasks by using the nanocomposites. Polymer-based nanofibers with a large surface area-to-volume ratio have shown great potential for use in facemasks to achieve both high filtration efficiency and sufficient air permeability.71–73 Yang et al.74 presented a design of the nanofiber-based facemasks for a better thermal comfort of the user. The facemask was made of hybrid nanocomposites containing electrospun nylon-6 nanofibers on top of the needle-punched nanoporous polyethylene (nanoPE) substrate. While nanofibers with strong particulate matter (PM) adhesion properties ensured high PM capture efficiency (99.6% for PM2.5) with a low pressure drop, a nanoPE substrate with high infrared (IR) transparency (92.1%, weighted based on human body radiation) resulted in effective radiative cooling. Figures 3(a)–3(c) show the schematic, photographs, and scanning electron micrographs of the proposed hybrid nanofiber-based facemask. The comparative PM capture efficiency and air permeability results have demonstrated the superiority of the presented facemask over the commercial masks [Figs. 3(d) and 3(e)]. Moreover, the thermal image revealed that the fiber/nanoPE facemasks had high transparency to the human body radiation (cooling effect). In contrast, the commercial facemasks blocked a large portion of it. They further modified the nanoPE substrate with Ag coating and demonstrated that fiber/Ag/nanoPE had a warming effect.  FIG. 3. [(a) and (b)] Schematics of the proposed hybrid facemask (nanofibers/nanoPE) and its photograph. (c) The SEM images show the condition of nylon-6 fibers before and after filtering the particulate matter (PM). [(d) and (e)] The removal efficiency of the fiber/nanoPE facemasks compared to two commercial masks, and their pressure drop spectra as a function of the wind velocity. Reproduced with permission from Yang et al., “Thermal management in nanofiber-based face mask,” Nano Lett. 17, 3506 (2017). Copyright 2017 American Chemical Society. Zhang et al.75 reported the use of an active ventilation fan to reduce the dead space temperature and CO2 level. An infrared camera (IRC) method was used to elucidate the temperature distribution on the prototype FFR’s outside surface and the wearer’s face, and surface temperature was found to be lowered notably. Both the inside and outside temperatures resulted from the simulation were found to be in good agreement with experimental results. However, the inward blowing fans may compromise the filtering effectiveness of the facemask. There are commercially available facemasks fitted with a one-way valve for facilitating the removal of humidity and expired air within the space between the facemask and the face. However, during the COVID-19 pandemic, one of the main reasons for wearing the mask is not only to protect the inhalation of the virus but also to prevent the spread of the virus into the air if the wearer happens to be a carrier of the virus. If the wearer is a healthy subject, the use of a one-way valve and ventilation fan would, indeed, mitigate the buildup of humidity and carbon dioxide within the dead space. Zhu et al.76 reported a three-dimensional model of a normal human nasal cavity to simulate the volume of fraction (VOF) of both fresh air and respired air within the nasal cavity. The model consisted of a large rectangular domain outside the nasal cavity representing ambient air, human nasal cavity, and partial of the pharynx. This was the first reported piece of work that modeled the details of nasal cavity instead of just the nostrils as openings for the flow simulations. The advantage of this simulation was that the flow field within the space between the nostrils and the facemask could be more accurately simulated as the boundary condition could be specified away from the nostril at the pharyngeal area. Two cases were simulated. Case I refers to a human face with a N95 respirator onto the human face, and case II refers to a human face without a respirator. The results showed that above 60% of inspired air was respired air in case I compared to less than 1.2% in case II. During expiration, the volume of fraction (VOF) of respired air in both the cases was above 95%. The streamlines at peak inspiration were relatively smooth while entering the cavity in both the cases; while at peak expiration, large vortex was observed within the air space between the human face and the respirator in case I. For future studies, one could explore the in vivo experimental studies with the use of miniaturized and wireless sensors for monitoring not just the temperature but also the humidity and carbon dioxide content within the space between the nostrils and the facemask. The sensors need to be small so as not to disrupt the flow fields. If a single sensor cannot be small enough for the measurement of all the three parameters, one may need to have separate sensors and repeat the experiment for the same human subject. B. Flow resistance Another important parameter affecting the comfort of the wears is the flow resistance of the facemask. In principle, if the flow resistance is lower while maintaining the same filtering efficiency, the comfort level will be enhanced. However, the facemask’s flow resistance is just an indicator and does not specify the wearer’s breathing resistance. While the flow resistance could be measured using a typical setup for correlating the fluid flow rate to the pressure drop across the facemasks, the breathing resistance could only be measured using a human subject or a replica of the nasal pharyngeal system. Lee and Wang77 presented the pioneering work of measuring the nasal airflow resistance during inspiration and expiration using a standard rhinomanometry and nasal spirometry. A modified full-facemask was produced in-house to measure nasal resistance using N95 (3M 8210) respirators. The results showed a mean increment of 126% and 122% in inspiratory and expiratory flow resistances, respectively, with N95 respirators. There was also an average reduction of 37% in air exchange volume with the use of N95 respirators. The same group did a follow-up study investigating the change in human nasal functions after wearing an N95 respirator and a surgical facemask.78 The human subject study involved 87 healthy healthcare workers. Each of the volunteers attended two sessions and wore an N95 respirator in session 1 (S1) and surgical facemask in session 2 (S2) for 3 h. The mean minimum cross-sectional area (mMCA) of the two nasal airways via acoustic rhinometry and nasal resistance via rhinomanometry was measured before and immediately after the mask. The equipment could not be used to perform in vivo measurement with the facemask on. Rhinomanometry was repeated every 30 min for 1.5 h after the removal of masks. A questionnaire was distributed to each of the volunteers during the 3 h mask-wearing period to report subjective feelings on the discomfort level of breathing activity. Among 77 volunteers who completed both the two sessions, the mean nasal resistance immediately increased upon removing the surgical facemask and N95 respirator. The mean nasal resistance was significantly higher in S1 than S2 at 0.5 h and 1.5 h after removing the masks (p < 0.01). There was an increase in nasal resistance upon the removal of the N95 respirator and surgical facemask potentially due to nasal physiological changes. The N95 respirator caused higher post-wearing nasal resistance than the surgical facemask with different recovering routines. This was the first time that the effect of long duration wearing of a facemask was objectively monitored. However, the duration of 3 h for wearing a facemask was deemed to be too short under the current COVID-19 simulations, and a human subject study for a longer duration of wearing a facemask should be attempted. The research could also be enhanced using miniaturized pressure, temperature, humidity, and gas sensors for in vivo monitoring of the air condition within the space between the nostrils and the facemask. Such experimental data would be useful for validating numerical models for assessing the comfort level for wearing different types of facemask. Another potential approach is to develop a replica for replacing a human subject for such a long duration study, similar to the use of an acoustic head for replacing human subjects in the more extended duration noise exposure study. Zhu et al.79 reported another investigation on the effect of long duration wearing of N95 and surgical facemasks on upper airway functions. A total of 47 volunteers of National University Hospital of Singapore participated for the study. Each of the volunteers wore both the N95 respirator and the surgical facemask for 3 h on two different days. During the period of mask wearing, relative airflow rates were recorded. The study revealed the increased level of discomfort to the user with time while wearing the masks. Moreover, the N95 respirator caused higher post-wearing nasal resistance than the surgical facemask with different recovering routines. |
|
