|
|
Post by Admin on Nov 20, 2020 21:03:08 GMT
Results and Discussion We examined SARS-CoV-2 infections and COVID-19 outcomes among patients who had previously been assessed with a comprehensive respiratory panel polymerase chain-reaction (CRP-PCR, FilmArray Respiratory Panel, RP2, BioFire Diagnostics) test. The CRP-PCR detects nucleic acids for the 4 eCoVs along with 16 other pathogens, and thus, a positive test indicates ongoing rather than prior infections. We retrospectively collected data from patients with an available CRP-PCR result from May 18, 2015 to March 11, 2020 in the electronic medical record (EMR). March 11, 2020 was chosen as the end date because the first available SARS-CoV-2 test in the Boston Medical Center (BMC) EMR was on March 12, 2020. We also obtained all SARS-CoV-2 reverse transcription PCR (RT-PCR) results between March 12, 2020 and June 12, 2020 that were available in the EMR. Analysis was restricted to patients not recorded deceased prior to March 11, 2020, above 18 years of age, and with the first SARS-CoV-2 result at least 7 days after the CRP-PCR test. 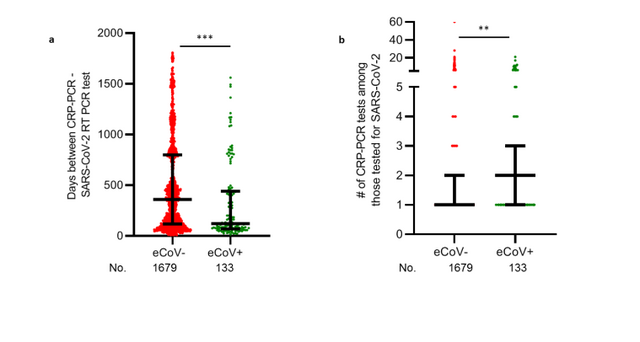 Fig. 1 A total of 15,928 different patients had at least one CRP-PCR test. An eCoV was previously detected in 875 of these patients (termed eCoV+), and the remaining 15,053 individuals (classified as eCoV-) never had a documented eCoV infection. Most but not all, of the demographic characteristics showed no significant difference between the eCoV+ and eCoV- group (Table 1), although there were some variation in race and human immunodeficiency virus (HIV) infection status. The proportion of patients with no, one, or two or more co-morbidities was not significantly different between the eCoV+ as compared to eCoV- group. The CRP-PCR test was more frequently ordered while patients were at a hospital (inpatient, observation unit, or emergency department) in the eCoV- as compared to eCoV+ patients. These observations imply that the patients in the two groups had relatively similar level of pre-existing morbidity, but the eCoV- as compared to the eCoV+ patients may have had more severe clinical presentation at the time of CRP-PCR testing. A total of 1,812 (11.4%) of the patients under investigation had an available SARS-CoV-2 result (Table 2). A significantly higher proportion of eCoV+ (15.2%) individuals were tested for SARS-CoV-2 as compared to the eCoV- (11.2%) patients (odds ratio (OR) 1.4, 95% confidence interval (CI) 1.2 – 1.7). The odds of SARSCoV-2 testing (OR 1.4, 95% CI 1.2 – 1.7) remained significantly higher in the eCoV+ as compared to eCoVpatients after adjusting for race/ethnicity, chronic obstructive pulmonary disease, HIV, number of comorbidities, and level of clinical care. The last documented CRP-PCR result prior to the SARS-CoV-2 RT-PCR test occurred significantly more recently in the eCoV+ (median 121 days, interquartile range (IQR) 69 - 440 days) as compared to the eCoV- (median 359 days, IQR 117 - 799 days, p < 0.0001) patients (Fig. 1a). The eCoV+ (median 2, IQR 1 - 3) as compared to eCoV- (median 1, IQR 1 – 2, p = 0.002) patients also had significantly more frequent CRP-PCR testing (Fig. 1b). The more recent and frequent CRP-PCR testing in the eCoV+ individuals suggests a greater likelihood of having a clinical presentation prompting respiratory evaluation. The greater likelihood of illness prompting CRP-PCR evaluation may also account for the higher level of SARS-CoV-2 RT-PCR testing among the eCoV+ as compared to eCoV-group. Among the patients evaluated for SARS-CoV-2, 470 (25.9%) had at least 1 positive SARS-CoV-2 RTPCR at some point (Table 2). A total of 252 (53.6%) of the SARS-CoV-2 infected patients had a COVID-19 related hospitalization during the study period. The frequency of documented SARS-CoV-2 infection among those tested, and of hospitalization among those infected, did not differ between the eCoV+ and eCoV- groups (Table 2). Some risk factors associated with more severe COVID-19, such as older age, male gender, higher body mass index (BMI), and pre-existing diabetes mellitus (DM) (9, 10), were significantly different between the eCoV+ as compared to eCoV- patients that were eventually hospitalized after SARS-CoV-2 infection (Supplemental Table 1). The number of prior diagnoses, however, were not different among the hospitalized eCoV+ and eCoV- group, suggesting they had a similar level of pre-existing morbidity. The eCoV+ as compared to eCoV- hospitalized patients had a significantly lower odds for intensive care unit (ICU) admission (OR 0.1, 95% CI 0.0 – 0.7) and a trend towards lower odds of mechanical ventilation (OR 0.0, 95% CI 0.0 – 1.0). The odds of ICU care (OR 0.1, 95% CI 0.1–0.9) remained significantly lower in the eCoV+ as compared to eCoV- patients after adjusting for age, gender, BMI, and DM. The percentage of hospitalized patients that eventually died over follow up was lower in the eCoV+ (4.8%) as compared to the eCoV- (17.7%) group. Survival probability was significantly higher in the hospitalized eCoV+ as compared to eCoV- COVID-19 hospitalized patients (hazard ratio (HR) 0.3, 95% CI 0.1 – 0.7, Fig. 2). After adjusting for age, gender, BMI, and DM, the HR remained 0.3, although the confidence interval became much wider and encompassed unity (0.0 – 2.0). Cumulatively, these observations suggest that recent documented eCoV infection is associated with less severe COVID-19. Lower virus levels in the respiratory tract associate with less severe COVID-19 (11). The EMR provides scant information from which a patient’s burden of infection may be inferred, but cycle threshold (Ct) values from SARS-CoV-2 tests may be used for extrapolation. Both, an Abbott commercial assay and an in-house assay (12) were used to determine the presence of SARS-CoV-2 in our hospital during this study period due to testing and material limitations. Patients tested with the two assays did not show significantly different Ct values (p = 0.13, Supplemental Fig. 1a). In the patient’s initial or only positive SARS-CoV-2 RT-PCR, the eCoV+ as compared to eCoV- patients had, if anything, more abundant virus in the nasopharynx as indicated by lower Ct values (Supplemental Fig. 1b). A multivariable linear regression model demonstrated that the eCoV+ as compared to the eCoV- patients had around a 4 unit lower Ct value (β = 4.0, 95% CI -0.6 – 8.7, p =0.09), but this difference was not statistically significant. The in-house assay also trended to yield around 2 unit lower Ct values compared to the commercial Abbott assay (β = 2.0, 95% CI -0.3 – 4.4, p = 0.09). In both analyses, the data did not reach statistical significance, and the number of data points was extremely limited especially for the eCoV+ patients. Although limited by the upper airway site of sampling, single time of analysis, and small sample size, these data do not support the hypothesis that the better outcome patients were due to lower viral burdens. Combined with the SARS-CoV-2 acquisition frequency data (Table 2), these observations potentially imply that the eCoV+ patients do not possess immunological memory that constrains initial virus replication. |
|
|
|
Post by Admin on Nov 20, 2020 22:14:47 GMT
Less severe outcomes from similar viral loads may be due to decreased “cytokine storm” or inflammatory injury. Higher plasma inflammatory markers, such as C-reactive protein (CRP) and lactate dehydrogenase (LDH), correlate with more severe disease (13), supporting the concept that inflammatory responses contribute to pathophysiology. The eCoV+ as compared to the eCoV- patients trended towards lower CRP (eCoV+ median 24.0 ng/L, IQR 7.2 – 69.3 ng/L versus eCoV- median 55.1 ng/L, IQR 16.6 – 109.0 ng/L, p = 0.06) and LDH (eCoV+ median 284.0 units/L, IQR 191 – 344.5 units/L versus eCoV- median 306.0 units/L, IQR 241 – 385.5 units/L, p = 0.09) levels upon their initial presentation for their COVID-19 related hospitalization (Supplemental Fig. 2a and 2b). These observations possibly suggest that patients with a previously documented eCoV infection may have more subdued inflammatory responses soon after SARSCoV-2 infection (14, 15). As a whole, patients with previous CRP-PCR tests prior to SARS-CoV-2 acquisition are hospitalized at a higher frequency as compared to the general population at our and other medical centers (16, 17). This population may represent a sicker group with a higher propensity to acquire a respiratory infection and require hospitalization. We find that, within that population, those with a recent prior documented eCoV infection were more likely to have a clinical presentation triggering SARS-CoV-2 testing, but their likelihood of being infected was relatively similar. The level of hospitalization after infection also did not differ between the two groups. We interpret these data to suggest that those with recent eCoV infection may not have neutralizing immunity that prevents acquisition. Indeed, a previous study suggests that neutralizing responses against eCoVs are relatively short lived, and previously infected patients are susceptible to re-infection albeit with less severe disease (18). Importantly, we observed that the eCoV+ as compared to the eCoV- group was less likely to have ICU admission and death after COVID-19 diagnosis. Even without neutralizing immunity, patients with prior eCoV infections may have lung-localized primed immune responses that prevent severe disease from a heterologous virus (19). Heterotypic lung-localized resident memory T and B cells prevent severe infections from respiratory pathogens (20). Future studies should determine whether lung-localized heterotypic immunity is elicited by prior eCoV infection and is capable of ameliorating COVID-19 manifestations. The durability and extent of the potential immune protection and distinct effects of different eCoVs will also need to be investigated. 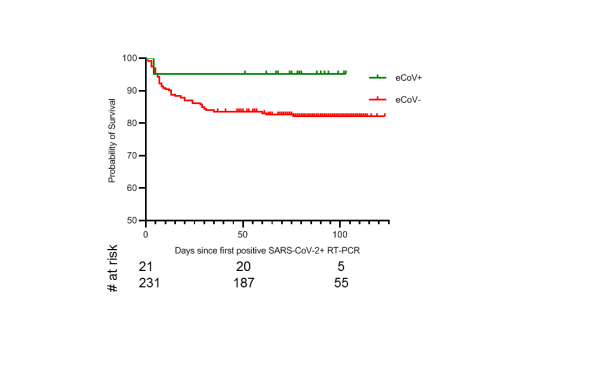 Fig. 2 This study had limitations. It was associative, and thus cannot determine causality. It involved small numbers from one hospital, so findings may not generalize. The observed morbidity and mortality may be linked to but not directly caused by SARS-CoV-2 infection. The absence of an eCoV+ result does not preclude coronavirus infections throughout the study period, so some individuals may have been classified inappropriately. The relatively low observed morbidity and mortality in the eCoV+ group, however, suggests that removing individuals with undocumented eCoV infection from the eCoV- group would further increase the effect size away from the null. Several different RT-PCR assays were used for SARS-CoV-2 testing at our institution during the study period; inherent differences in their targets and Ct values are acknowledged. With these limitations, results suggest prior eCoV infection was associated with less severe COVID-19. Larger studies and causal investigations are needed to identify the mechanisms and persistence of this inferred heterotypic immune protection. |
|
|
|
Post by Admin on Sept 7, 2021 19:45:06 GMT
Some scientists have called it "superhuman immunity" or "bulletproof." But immunologist Shane Crotty prefers "hybrid immunity." "Overall, hybrid immunity to SARS-CoV-2 appears to be impressively potent," Crotty wrote in commentary in Science back in June. www.science.org/doi/abs/10.1126/science.abj2258No matter what you call it, this type of immunity offers much-needed good news in what seems like an endless array of bad news regarding COVID-19. Over the past several months, a series of studies has found that some people mount an extraordinarily powerful immune response against SARS-CoV-2, the coronavirus that causes the disease COVID-19. Their bodies produce very high levels of antibodies, but they also make antibodies with great flexibility — likely capable of fighting off the coronavirus variants circulating in the world but also likely effective against variants that may emerge in the future. "One could reasonably predict that these people will be quite well protected against most — and perhaps all of — the SARS-CoV-2 variants that we are likely to see in the foreseeable future," says Paul Bieniasz, a virologist at Rockefeller University who helped lead several of the studies.  In a study published online last month In a study published online last month, Bieniasz and his colleagues found antibodies in these individuals that can strongly neutralize the six variants of concern tested, including delta and beta, as well as several other viruses related to SARS-CoV-2, including one in bats, two in pangolins and the one that caused the first coronavirus pandemic, SARS-CoV-1. "This is being a bit more speculative, but I would also suspect that they would have some degree of protection against the SARS-like viruses that have yet to infect humans," Bieniasz says. So who is capable of mounting this "superhuman" or "hybrid" immune response? People who have had a "hybrid" exposure to the virus. Specifically, they were infected with the coronavirus in 2020 and then immunized with mRNA vaccines this year. "Those people have amazing responses to the vaccine," says virologist Theodora Hatziioannou at Rockefeller University, who also helped lead several of the studies. "I think they are in the best position to fight the virus. The antibodies in these people's blood can even neutralize SARS-CoV-1, the first coronavirus, which emerged 20 years ago. That virus is very, very different from SARS-CoV-2." In fact, these antibodies were even able to deactivate a virus engineered, on purpose, to be highly resistant to neutralization. This virus contained 20 mutations that are known to prevent SARS-CoV-2 antibodies from binding to it. Antibodies from people who were only vaccinated or who only had prior coronavirus infections were essentially useless against this mutant virus. But antibodies in people with the "hybrid immunity" could neutralize it. These findings show how powerful the mRNA vaccines can be in people with prior exposure to SARS-CoV-2, she says. "There's a lot of research now focused on finding a pan-coronavirus vaccine that would protect against all future variants. Our findings tell you that we already have it. "But there's a catch, right?" she adds: You first need to be sick with COVID-19. "After natural infections, the antibodies seem to evolve and become not only more potent but also broader. They become more resistant to mutations within the [virus]." Hatziioannou and colleagues don't know if everyone who has had COVID-19 and then an mRNA vaccine will have such a remarkable immune response. "We've only studied the phenomena with a few patients because it's extremely laborious and difficult research to do," she says. But she suspects it's quite common. "With every single one of the patients we studied, we saw the same thing." The study reports data on 14 patients. Several other studies support her hypothesis — and buttress the idea that exposure to both a coronavirus and an mRNA vaccine triggers an exceptionally powerful immune response. In one study, published last month in The New England Journal of Medicine, scientists analyzed antibodies generated by people who had been infected with the original SARS virus — SARS-CoV-1 — back in 2002 or 2003 and who then received an mRNA vaccine this year. Remarkably, these people also produced high levels of antibodies and — it's worth reiterating this point from a few paragraphs above — antibodies that could neutralize a whole range of variants and SARS-like viruses. Now, of course, there are so many remaining questions. For example, what if you catch COVID-19 after you're vaccinated? Or can a person who hasn't been infected with the coronavirus mount a "superhuman" response if the person receives a third dose of a vaccine as a booster? Hatziioannou says she can't answer either of those questions yet. "I'm pretty certain that a third shot will help a person's antibodies evolve even further, and perhaps they will acquire some breadth [or flexibility], but whether they will ever manage to get the breadth that you see following natural infection, that's unclear." Immunologist John Wherry, at the University of Pennsylvania, is a bit more hopeful. "In our research, we already see some of this antibody evolution happening in people who are just vaccinated," he says, "although it probably happens faster in people who have been infected." In a recent study, published online in late August, Wherry and his colleagues showed that, over time, people who have had only two doses of the vaccine (and no prior infection) start to make more flexible antibodies — antibodies that can better recognize many of the variants of concern. So a third dose of the vaccine would presumably give those antibodies a boost and push the evolution of the antibodies further, Wherry says. So a person will be better equipped to fight off whatever variant the virus puts out there next. "Based on all these findings, it looks like the immune system is eventually going to have the edge over this virus," says Bieniasz, of Rockefeller University. "And if we're lucky, SARS-CoV-2 will eventually fall into that category of viruses that gives us only a mild cold." |
|
|
|
Post by Admin on Sept 7, 2021 22:48:19 GMT
Immunity to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is a vital issue for global society. Determining the quality and duration of that immunity is therefore key. But the adaptive immune system is complex, and these factors may differ between natural immunity (obtained by infection) and vaccine-generated immunity (1). Additionally, there is the question of the combination: What kind of immunity develops in people with natural immunity who are subsequently vaccinated? Such “hybrid immunity” is particularly interesting because of the notable finding that people with previous SARS-CoV-2 infection mount unusually potent immune responses to COVID-19 vaccines (2). This is exemplified in two studies in this issue on pages 1413 and 1418 by Stamatatos et al. (3) and Reynolds et al. (4), respectively, which also highlight natural and vaccine-induced immune responses to variants. Immunological memory is the source of protective immunity. Natural immunity and vaccine-generated immunity to SARS-CoV-2 are two different paths to protection. The adaptive immune system consists of three major branches: B cells (the source of antibodies), CD4+ T cells, and CD8+ T cells. For natural immunity, immunological memory to SARS-CoV-2 has been observed for more than 8 months for CD4+ T cells, CD8+ T cells, memory B cells, and antibodies (5), with a relatively gradual decline that appears to partially stabilize within a year (5–7). Levels of immunity can be placed on a spectrum, and natural immunity against symptomatic infection (COVID-19) has been found to be between 93 and 100% over 7 to 8 months in large studies, including locations where the SARS-CoV-2 variant of concern (VOC) B.1.1.7 (alpha) was widespread. Natural immunity against variants with changes that substantially reduce antibody recognition [e.g., B.1.351 (beta), P.1 (gamma), B.1.526 (iota), and B.1.617] is less clear; there is evidence of more reinfections with such variants (8). Neutralizing antibody activity against most VOCs is reduced for natural immunity and vaccine-generated immunity. That most VOCs have mutations engendering partial antibody escape is evidence of selection pressure to evade natural immunity. The biological relevance of the reductions in neutralizing antibody potency against variants is most clearly evident from vaccine clinical trials and observational studies. Among current COVID-19 vaccines in use, ChAdOx1 nCoV-19 (AstraZeneca) vaccine efficacy against symptomatic cases dropped from 75% to 11% against B.1.351 (9). By contrast, BNT162b2 (Pfizer/BioNTech) vaccine efficacy against symptomatic cases dropped from ∼95% to 75% against B.1.351, and protection against severe disease remained at 97% (10). Initial reports suggest that both vaccines retain most of their efficacy against B.1.617.2 (delta).  What happens when previously infected individuals are vaccinated? The observations in several studies, including those by Stamatatos et al. and Reynolds et al., are that an impressive synergy occurs—a “hybrid vigor immunity” resulting from a combination of natural immunity and vaccine-generated immunity (see the figure). When natural immunity to SARS-CoV-2 is combined with vaccine-generated immunity, a larger-than-expected immune response arises. There appear to be both B cell and T cell components to hybrid immunity. An important question about antibody-mediated immunity against VOCs has been whether neutralizing antibody reductions are due to intrinsically low antigenicity of the VOCs. That is, is it intrinsically challenging for B cells to recognize the variants' mutated spike proteins? The answer is no. Studies of natural infection with B.1.351 showed that neutralizing antibody responses were robust against that variant and the ancestral strain (11). Moreover, neutralizing antibodies against B.1.351 after vaccination of individuals previously infected with non-B.1.351 SARS-CoV-2 were ∼100 times higher than after infection alone and 25 times higher than after vaccination alone—even though neither the vaccine nor infection involved the B.1.351 spike. This enhanced neutralizing breadth was first reported by Stamatatos et al. and then confirmed by multiple groups (4, 12). Overall, the strength and breadth of the antibody responses after vaccination of previously SARS-CoV-2–infected persons was unanticipated. Why does this pronounced neutralizing breadth occur? Memory B cells are a primary reason. They have two major functions: one is to produce identical antibodies upon reinfection with the same virus, and the other is to encode a library of antibody mutations, a stockpile of immunological variants. These diverse memory B cells, created in response to the original infection, appear to be preemptive guesses by the immune system as to what viral variants may emerge in the future. This brilliant evolutionary strategy is observed clearly for immunity to SARS-CoV-2: A substantial proportion of memory B cells encode antibodies that are capable of binding or neutralizing VOCs, and the quality of those memory B cells increases over time (7). Thus, the increase in variant-neutralizing antibodies after vaccination of previously SARS-CoV-2–infected persons reflects recall of diverse and high-quality memory B cells generated after the original infection (7, 12). T cells are required for the generation of diverse memory B cells. The evolution of B cells in response to infection, or vaccination, is powered by immunological microanatomical structures called germinal centers, which are T cell–dependent, instructed by T follicular helper (TFH) CD4+ T cells. Thus, T cells and B cells work together to generate antibody breadth against variants. Additionally, T cells appear to be important at the recall stage. Memory B cells do not actively produce antibodies; they are quiescent cells that only synthesize antibodies upon reinfection or subsequent vaccination. Memory B cells are increased 5- to 10-fold in hybrid immunity compared with natural infection or vaccination alone (3, 12). Virus-specific CD4+ T cells and TFH cells appear to be key drivers of the recall and expansion of those SARS-CoV-2 memory B cells and the impressive antibody titers observed (13, 14). Antibodies are clearly involved in protection against SARS-CoV-2 reinfection, but evidence also points to contributions from T cells (1). T cell responses against SARS-CoV-2 in natural infection are quite broad (1), and most T cell epitopes are not mutated in VOCs, indicating that the contributions of T cells to protective immunity are likely to be retained (4, 15). Most of the COVID-19 vaccines in use consist of a single antigen, spike, whereas 25 different viral proteins are present in SARS-CoV-2. Thus, the epitope breadth of both the CD4+ and CD8+ T cell responses is more restricted in current COVID-19 vaccines than in natural infection (1), whereas hybrid immunity consists of both spike and non-spike T cell memory. Notably, the Pfizer/BioNTech and Moderna COVID-19 messenger RNA (mRNA) vaccines can substantially boost spike CD4+ T cell responses in previously infected persons after one immunization (3, 4, 13, 14). Differences in T cell responses after two doses of vaccine are more variable in those individuals (3, 13). The immune system treats any new exposure—be it infection or vaccination—with a cost-benefit threat analysis for the magnitude of immunological memory to generate and maintain. There are resource-commitment decisions: more cells and more protein throughout the body, potentially for decades. Although all of the calculus involved in these immunological cost-benefit analyses is not understood, a long-standing rule of thumb is that repeated exposures are recognized as an increased threat. Hence the success of vaccine regimens split into two or three immunizations. Heightened response to repeated exposure is clearly at play in hybrid immunity, but it is not so simple, because the magnitude of the response to the second exposure (vaccination after infection) was much larger than after the second dose of vaccine in uninfected individuals. Additionally, the response to the second vaccine dose was minimal for previously infected persons, indicating an immunity plateau that is not simple to predict. Moreover, previously infected people in some SARS-CoV-2 vaccine studies included both asymptomatic and symptomatic COVID-19 cases. Enhanced vaccine immune responses were observed in both groups, indicating that the magnitude of hybrid immunity is not directly proportional to previous COVID-19 severity. Overall, hybrid immunity to SARS-CoV-2 appears to be impressively potent. The synergy is primarily observed for the antibody response more so than the T cell response after vaccination, although the enhanced antibody response depends on memory T cells. This discordance needs to be better understood. Will hybrid natural/vaccine-immunity approaches be a reproducible way to enhance immunity? The Shingrix vaccine to prevent shingles, which is given to people previously infected with the varicella zoster virus, is impressively effective (∼97% efficacy), and elicits much higher antibody responses than viral infection alone. These principles also apply to combinations of vaccine modalities. It has long been observed that combining two different kinds of vaccines in a heterologous prime-boost regimen can elicit substantially stronger immune responses than either modality alone—depending on the order in which they are used and on which vaccine modalities are combined—for reasons that are not well understood. This may occur with combinations of COVID-19 vaccines, such as mRNA and adenoviral vectors, or mRNA and recombinant protein vaccines. These recent findings about SARS-CoV-2 immunology are pleasant surprises and can potentially be leveraged to generate better immunity to COVID-19 and other diseases. |
|
|
|
Post by Admin on Sept 8, 2021 21:10:07 GMT
Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant List of authors. Jamie Lopez Bernal, F.F.P.H., Ph.D., Nick Andrews, Ph.D., Charlotte Gower, D.Phil., Eileen Gallagher, Ph.D., et al.  Abstract BACKGROUND The B.1.617.2 (delta) variant of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes coronavirus disease 2019 (Covid-19), has contributed to a surge in cases in India and has now been detected across the globe, including a notable increase in cases in the United Kingdom. The effectiveness of the BNT162b2 and ChAdOx1 nCoV-19 vaccines against this variant has been unclear. METHODS We used a test-negative case–control design to estimate the effectiveness of vaccination against symptomatic disease caused by the delta variant or the predominant strain (B.1.1.7, or alpha variant) over the period that the delta variant began circulating. Variants were identified with the use of sequencing and on the basis of the spike (S) gene status. Data on all symptomatic sequenced cases of Covid-19 in England were used to estimate the proportion of cases with either variant according to the patients’ vaccination status. RESULTS Effectiveness after one dose of vaccine (BNT162b2 or ChAdOx1 nCoV-19) was notably lower among persons with the delta variant (30.7%; 95% confidence interval [CI], 25.2 to 35.7) than among those with the alpha variant (48.7%; 95% CI, 45.5 to 51.7); the results were similar for both vaccines. With the BNT162b2 vaccine, the effectiveness of two doses was 93.7% (95% CI, 91.6 to 95.3) among persons with the alpha variant and 88.0% (95% CI, 85.3 to 90.1) among those with the delta variant. With the ChAdOx1 nCoV-19 vaccine, the effectiveness of two doses was 74.5% (95% CI, 68.4 to 79.4) among persons with the alpha variant and 67.0% (95% CI, 61.3 to 71.8) among those with the delta variant. CONCLUSIONS Only modest differences in vaccine effectiveness were noted with the delta variant as compared with the alpha variant after the receipt of two vaccine doses. Absolute differences in vaccine effectiveness were more marked after the receipt of the first dose. This finding would support efforts to maximize vaccine uptake with two doses among vulnerable populations. (Funded by Public Health England.) |
|






