|
|
Post by Admin on Jul 26, 2020 23:58:07 GMT
In the ongoing search for genes underlying social behavior, none has sparked more curiosity, if not controversy, than the gene that codes for monoamine oxidase A — MAOA. Nicknamed the “warrior gene,” a variant of the MAOA drew international attention nearly a decade ago when geneticist Rod Lea reported1 that it was more common in Maori — the indigenous Polynesians of New Zealand — than in whites. According to one journalist, Lea suggested this gene might be the source of poor health and increased rates of violent crime in Maori. Since the 1990s scientists have identified several versions of the MAOA, which are usually categorized as low-activity or high-activity variants. MAOA genes are classified based on how many times a short sequence — a functional strip of DNA — repeats itself within a variable region of the gene . The most common variant, MAOA-4R, has four repeats and is associated with high-activity breakdown of neurotransmitters. Alternate forms of the MAOA, including the 2-repeat (2R) and 3-repeat (3R) versions, contain fewer repeat sequences. The 2R and 3R variants are often lumped together in studies of the low-activity MAOA gene. (Although the 5R version has a large number of repeats, it too is less active than the 4R version.) The two classes of MAOA versions correlate with different behavioral tendencies. Low-activity variants are thought to lead to reduced levels of MAOA in the brain, possibly shifting mood by changing serotonin levels. 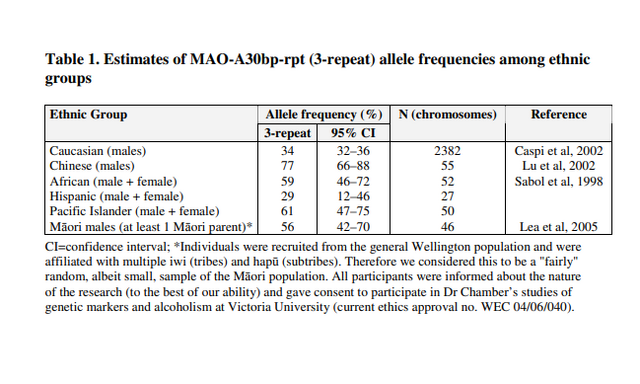 Historically, warfare was a central part of traditional Maori culture because, after all, these South Pacific islanders had to compete vigorously for limited natural resources. Today, some Maori are integrated into New Zealand society. Yet, overall they still lag behind other ethnic groups in their country in income, education, and health, and crime rates are higher. For many experts, this ethnic gap is the result of numerous environmental causes, including poverty. In 2006, Lea reported that MAOA-3R — one of the low-activity risky variants — was more common in Maori males than in white males. According to Lea, the 3R version was associated with a lineup of undesirable personality traits: risk-taking, violence, aggression, gambling, addiction and criminal behavior. MAOA-3R — the “original warrior gene” — was the first gene linked with antisocial characteristics. But Maori were not the only ethnic group with a high frequency of this variant. It turned out that while 3R was found in 56% of Maori males, it occurred in 58% of African American males and 34% of European males. Misinterpreted by the media, the 3R variant quickly became a lead character in a pop science narrative intended to explain why certain racial groups appear to have increased tendencies toward violence. When a disproportionately high number of males of an ethnic group carries a less common gene linked with aggressive behaviors, the discussion about that gene immediately takes on racial overtones. (Interestingly, the press ignored studies indicating that the 3R variant occurred in 61% of Taiwanese males and 56% of Chinese males). The relationship between the rare MAOA version and antisocial behaviors has raised eyebrows because, quite simply, this gene is not distributed equally across ethnic groups. In the Add Health database, 5.5% of African American men, 0.9% of Caucasian men, and 0.00067% of Asian men have 2R. (No information is currently available on the frequency of 2R in males of African black descent outside the United States.) Since the rare MAOA variant is virtually non-existent in whites, all of the males in Beaver’s study3 were black Americans. 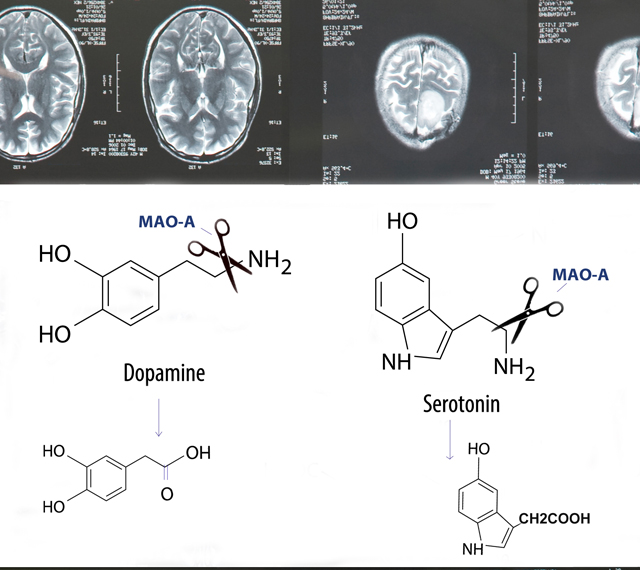 Beaver’s sample of 133 African American men from the Add Health database included 6% that carried 2R. Overall, 5.6% of the men in the sample reported shooting or stabbing someone at some point in their lifetime. The association between 2R and committing a shooting or stabbing crime was statistically significant. Based on Beaver’s evidence, 2R appears to increase the risk of shooting or stabbing a victim during adolescence or adulthood. For some commentators in the public arena, MAOA-2R has become a symbol of a new era in behavioral genetics research — an era that has reintroduced race into the nature versus nurture debate over the source of ethnic behavioral differences. It is not clear if MAOA-2R is an exception. The more common low-activity variant, 3R, interacts with adverse social effects such as childhood maltreatment. But other possible environmental factors, which conceivably could interact with the 2R, may not have been explored in-depth as yet. One such environmental influence that has recently received attention is parents and caregivers’ punitive discipline — spanking and yelling — of a young child. Punitive practices are not necessarily abuse. But in families that traditionally use harsh discipline with their children, corporeal punishment or even loud verbal chastising can at times turn into maltreatment.  Choe’s study3 is the first to demonstrate that ethnic minority children— African Americans, not just Caucasians — with a low-expression MAOA gene variant who face harsh discipline have an increased risk for antisocial behavior. Choe’s team published the effects of just the 3R variant, excluding five African American participants in their study carrying the 2R version. Curious about possibly different effects of 2R, they then reanalyzed the data to include the five black males with 2R. The findings remained the same. Combining the boys with 2R — the highest risk variant — and those with the less severe risky 3R did not change the differences the researchers found between the 3R and 4R variants. The five males with 2R comprised a very small sample, but the fact that both low-activity MAOA variants, 2R and 3R, interacted with an environmental factor — punitive discipline — at specific ages, or developmental milestones, is noteworthy. It suggests the effects of MAOA-2R on antisocial behaviors are partly mediated by non-genetic factors. Choe stresses that genetic influences on social behaviors such as juvenile delinquency cannot be fully understood outside the context of social circumstances. He is referring not only to parenting styles, but also to the in utero environment of the unborn fetus. As he explains, compared to the white youth in his study, the African Americans were more likely to grow up in poorer, urban, dangerous neighborhoods. A high percentage of these youth are being raised by single mothers, and they grow up without the attention found in most middle-class homes. Choe acknowledges the role of genes in behavior, but he clearly thinks that environmental factors contribute substantially to ethnic differences in antisocial behaviors. As he points out, the white kids in the sample were also poor, but they lived in low-income suburban communities, not in densely concentrated inner cities. The suburbs pose less of a risk than urban communities for group delinquent behavior. 1. Lea R, Chambers G. Monoamine oxidase, addiction, and the “warrior” gene hypothesis. N Z Med J. 2007. 120 (1250) PMID: 17339897. 2. Beaver K, Barnes J, Boutwell B. The 2-repeat allele of the MAOA gene confers an increased risk for shooting and stabbing behaviors. Psychiatr Q. 2013a. Dec 11. 3. Choe D, Shaw D, Hyde L, Forbes E. Interactions between monoamine oxidase A and punitive discipline in African American and Caucasian men’s antisocial behavior. Clinical Psychological Science. 2014. March 14. doi:10.1177/2167702613518046 |
|
|
|
Post by Admin on Jul 31, 2020 0:04:03 GMT
IS Australia more likely to breed criminals as a result of its historically penal gene pool? Two variations of a gene have been found to play an important role in criminal behaviour.  Labelled a ‘warrior gene’ a study published in Molecular Psychiatry has found a link between two gene variations and a person’s inclination toward violent crime. The study found a variation in genes that, when activated, produced a different brain chemical reaction to other people. It estimated around five to 10 per cent of all severe violent crime in Finland could be attributed to the genotype. While scientists warn genes alone don’t automatically create a criminal, their existence could have genetic ramifications for a nation historically populated by a mass migration of criminals. British neuroscientist Prof Adrian Raine was one of the first to do brain scans on murderers. He found reduced activity in the prefrontal cortex — which controls emotional impulse — and greater activity in the amygdala — which generates emotion. Raine’s study revealed that childhood abuse that caused damage to the front of the head could result in more rage and anger. And in In his book The Anatomy of Violence he looks at how genetics and environmental influences can work to create a criminal. But professor in Psychiatry at the University of California, Jim Fallon, is proof that not all bad genes lead to a life of crime. He told the BBC he became interested in genetics after discovering a large quantity of murderers in his family tree. “People with far less dangerous genetics become killers and are psychopaths than what I have. I have almost all of them,” he told BBC Magazine. “If you’ve the high-risk form of the gene and you were abused early on in life, your chances of a life of crime are much higher,” he said. “If you have the high-risk gene but you weren’t abused, then there really wasn’t much risk. So just a gene by itself, the variant doesn’t really dramatically affect behaviour, but under certain environmental conditions there is a big difference.”  So is Australia a pool of deactivated criminal minds awaiting a perfect storm? “Our population may have exhibited more independent nonconformist survival behaviour, a strong sense of justice, good problem solving, a tendency not to follow the leader, a strong sense of humour and so on,” Dr Bambling said. “It would make sense that all the nonconformists would have been shipped out, and this nonconformity has become part of the Australian culture.” But the good news is, we’re probably in control of the urge to break the rules.  “The best research suggests that in most cases we are well able to override our behavioural genetics with our minds,” Dr Bambling said. “If we build up good self-regulation skills in childhood or even learn them later in life we can manage even our most volatile genetic behavioural tendencies.” “Environment is most important for most cases. For some people who have all the genetic and social risk factors together, prospects are not bright. “For the warrior gene it may not express in the same way if a person grew up in a healthy environment, and in this way environment again is the trigger either way,” Dr Bambling said. “Adults can develop impulse control with psychological training, even if they had no control beforehand. “The idea of one gene and one action is flawed. Our genetics perform mind-boggling actions and interactions of great complexity and there is much to be learned.” Genetic background of extreme violent behavior J Tiihonen, M-R Rautiainen, H M Ollila, E Repo-Tiihonen, M Virkkunen, A Palotie, O Pietiläinen, K Kristiansson, M Joukamaa, H Lauerma, J Saarela, S Tyni, H Vartiainen, J Paananen, D Goldman & T Paunio Molecular Psychiatry volume 20, 786–792(2015) Abstract In developed countries, the majority of all violent crime is committed by a small group of antisocial recidivistic offenders, but no genes have been shown to contribute to recidivistic violent offending or severe violent behavior, such as homicide. Our results, from two independent cohorts of Finnish prisoners, revealed that a monoamine oxidase A (MAOA) low-activity genotype (contributing to low dopamine turnover rate) as well as the CDH13 gene (coding for neuronal membrane adhesion protein) are associated with extremely violent behavior (at least 10 committed homicides, attempted homicides or batteries). No substantial signal was observed for either MAOA or CDH13 among non-violent offenders, indicating that findings were specific for violent offending, and not largely attributable to substance abuse or antisocial personality disorder. These results indicate both low monoamine metabolism and neuronal membrane dysfunction as plausible factors in the etiology of extreme criminal violent behavior, and imply that at least about 5–10% of all severe violent crime in Finland is attributable to the aforementioned MAOA and CDH13 genotypes. |
|
|
|
Post by Admin on Aug 1, 2020 6:55:32 GMT
Genetic background of extreme violent behavior
J Tiihonen, M-R Rautiainen, H M Ollila, E Repo-Tiihonen, M Virkkunen, A Palotie, O Pietiläinen, K Kristiansson, M Joukamaa, H Lauerma, J Saarela, S Tyni, H Vartiainen, J Paananen, D Goldman & T Paunio
Molecular Psychiatry volume 20, 786–792(2015)
Abstract
In developed countries, the majority of all violent crime is committed by a small group of antisocial recidivistic offenders, but no genes have been shown to contribute to recidivistic violent offending or severe violent behavior, such as homicide. Our results, from two independent cohorts of Finnish prisoners, revealed that a monoamine oxidase A (MAOA) low-activity genotype (contributing to low dopamine turnover rate) as well as the CDH13 gene (coding for neuronal membrane adhesion protein) are associated with extremely violent behavior (at least 10 committed homicides, attempted homicides or batteries). No substantial signal was observed for either MAOA or CDH13 among non-violent offenders, indicating that findings were specific for violent offending, and not largely attributable to substance abuse or antisocial personality disorder. These results indicate both low monoamine metabolism and neuronal membrane dysfunction as plausible factors in the etiology of extreme criminal violent behavior, and imply that at least about 5–10% of all severe violent crime in Finland is attributable to the aforementioned MAOA and CDH13 genotypes.
Introduction
Violent crime is a major issue that affects the quality of life even in stable and wealthy societies. In industrialized countries, the majority of all violent crime is committed by a relatively small group of antisocial recidivistic offenders,1,2 and more than 50% of severe antisocial behavior is attributable to genetic factors.3 The classic study by Mednick et al.,4 reported a significant correlation between adoptees and their biological parents for property crimes, but not for violent crimes. However, a recent study using an enormous Swedish nationwide adoption database with a long follow-up period found convincing evidence that the criminal records of biological parents predicted both violent and non-violent criminality among their adopted away children.5 Two decades ago, it was observed that a rare mutation leading to a complete deficiency of monoamine oxidase A (MAOA) was associated with impulsive and aggressive behavior in a Dutch kindred.6 Thus far, only two studies have reported an association between a specific gene and criminal violent offending.7,8 In the study by Caspi et al.,7 55 (12%) of the boys who were studied had a combination of the low-activity MAOA promoter genotype and childhood maltreatment, which accounted for 44% of the violent convictions in their cohort.7 Although this finding has not been replicated, and the majority of violent convictions in this cohort were not severe, such as homicide or attempted homicide, this MAOA variant has become widely called as a ‘warrior gene’.
Recently, it was reported that this finding has actually started to influence the attitudes on court sentences in the US.9 A meta-analysis that included 11 000 individuals showed a significant interaction between the low-activity MAOA genotype and childhood adversities on a subsequent antisocial outcome.10 However, the largest study on this issue, with more than 4000 individuals, could not confirm the hypothesis that this MAOA genotype moderates the relationship between childhood maltreatment and antisocial behavior, but found statistically non-significant evidence for a main effect of MAOA genotype on having disposition toward violence.11 Thus, the issue of a ‘warrior gene’ has remained controversial.
In the study by Bevilacqua et al.,8 a stop codon in HTR2B was associated with substance abuse and a risk of committing impulsive crimes such as homicide, batteries and arsons, and impulsive behavior in mice. This finding indicated that HTR2B has a role in impulsivity, but it was not possible to conclude whether the functional variant was associated with substance abuse or violent behavior per se. In conclusion, no genes nor their polymorphisms have been shown to explain severe violent behavior in humans, nor contribute to recidivistic violent offending. Therefore, we conducted a candidate gene study on both MAOA and HTR2B, and performed a genome-wide association study (GWAS) in cohorts of Finnish violent offenders compared with the general population.
CRIME (Discovery cohort)
To obtain a comprehensive and representative cohort of Finnish offenders who had committed several severe violent crimes, we conducted a survey in the 19 largest prisons in Finland to identify such individuals. By using the National Prison Register, we obtained information on all prisoners serving their sentence in these prisons. We specifically asked for participation from those prisoners with Finnish origin who had committed at least two crimes that led to prison sentences as indications of an antisocial lifestyle. A total of 1004 prisoners were screened, of which, 184 (18.4%) refused to participate in the study and 26 (2.6%) were excluded because of a psychosis diagnosis. Altogether, the CRIME cohort included 794 prisoners. Those individuals who had committed only non-violent crimes were classified as non-violent offenders (n=215). Those with at least one violent crime conviction were classified as violent offenders (n=538) and those having committed at least 10 violent crimes were classified as extremely violent offenders (n=84). The subgroup of extremely violent offenders overlapped completely with the violent offenders.
Violent crime status was unknown for 41 individuals who were excluded from the cohort. Violent offences included murder, attempted murder, manslaughter, attempted manslaughter, other types of homicide and battery. Non-violent crimes were typically drunk-driving, drug-related crimes or property crimes. Offenders having committed only sexual crimes were excluded. The participants were interviewed with Structured Clinical Interview for DSM-IV- Disorders (SCID) to exclude individuals with a psychosis diagnosis, and to assess whether or not the subject fulfilled criteria for antisocial personality disorder. Also, any history of substance abuse (alcohol, heroin, buprenorphine, amphetamine, cannabis, other) was obtained through a questionnaire, which included also questions about maltreatment during childhood (e.g., good circumstances, indifferent parents or severe maltreatment such as family violence). The history of criminal convictions was obtained from the National Crime Register.
The subjects provided a written informed consent. This study was approved by the Ethics Committee for Pediatrics, Adolescent Medicine and Psychiatry, Hospital District of Helsinki and Uusimaa, and Criminal Sanctions Agency. All the subjects who participated in the study received a voucher of 20 euros for their participation.
Health 2000 control cohort
The Health 2000 study (http://www.terveys2000.fi/doc/methodologyrep.pdf) is a Finnish nationwide survey collected in 2000–2001. The study subjects were selected from the Social Insurance Institution (SII) of Finland (Kela) based on the assumption that these individuals would reflect the main demographic distributions of the Finnish population.
The main aims of this survey were to characterize the public health problems in Finland, in terms of both physical and mental health and work-related traits. The study consisted of home interviews and a health examination that included a laboratory examination conducted at a local health center. Blood samples for DNA extraction were also collected. In total, Health 2000 included 8028 individuals from the general population who were over 30 years of age (54% were female). Of these, 2124 (26%) belonged to a GenMets sub-cohort who were genotyped by whole-genome chips. Originally, the GenMets sample was collected to study metabolic syndrome, where half of the individuals had metabolic syndrome and the other half were age- and gender-matched controls.12 A written informed consent was obtained from participants. This part of the study was approved by the ethics committee of the Helsinki University Central Hospital.
In the present study, the Genmets subpopulation (n=2124) was used as a control group in the GWAS analyses. Following the discovery analyses and regenotyping, the entire Health 2000 cohort (n=6600) was further used as a study cohort in the analyses of candidate single nucleotide polymorphisms (SNPs) for the general population.
FINRISK and Corogene control cohorts
The National FINRISK surveys have been conducted in Finland every 5 years since 1972. The first study included population-based samples from the provinces of North Karelia and Kuopio. This survey targeted persons between 30 and 59 years of age and focused on cardiovascular risk factors in the Finnish population. Since then, five more areas have been included, that is, Helsinki and Vantaa, Turku and Loimaa, North Savo, Oulu, and Lappland, and the age limits were expanded to 25–74 years (http://www.ktl.fi/finriski).
The Corogene study consists of 5000 consecutive Finnish patients (N=5000) who were assigned to coronary angiogram by the Helsinki University Central Hospital from June 2006 to March 2008. Since then, a new cohort of the same size has been collected in 5-year intervals over a period of 12 months for each interval. A total of 2500 patients from the first Corogene cohort, as well as 2500 age-, geographical area- and sex-matched controls from the FINRISK surveys were selected for GWASs, and these were used as controls in the genome-wide association analysis of this study.13
Homicide offender cohort
The replication cohort included 114 violent offenders who had committed at least one homicide, and who underwent forensic mental examination because of the extreme nature of their crimes. All these offenders were males. The DNA samples were collected between 1987 and 1998, as a part of the Finnish-American studies (with NIAAA/NIH) (Behavioral, Biochemical, Endocrine and Genetic Study of Alcohol Abusing Violent Offenders). The violent offenders were studied as inpatients at the University Hospital of Helsinki.
Written informed consent was obtained from each participant. Protocols were approved by the Institutional Review Board (IBR) of the US National Institute of Health (NIH) and the National Institute of Mental Health (NIMH), and also by the Office for Protection from Research Risks (OPRR), the University of Helsinki Department of Psychiatry IRB, the University of Helsinki Central Hospital IRB, the Finnish Ministry of Social Affairs and Health and the Ethics Committee of the National Public Health Institute of Finland.
|
|
|
|
Post by Admin on Aug 2, 2020 7:13:43 GMT
Phenotypes Violent offending Violent offending was defined by counting those individuals with at least one sentence for any violent offence, based on data from the National Crime Register. Extremely violent offending The characterization of ‘extremely violent offending’ was obtained by first calculating percentiles from the quantitative trait of ‘violent offending’ for the entire sample of criminals, and then dichotomizing that by using the 90th percentile cutoff value in the GWAS (i.e., those convicted of 10 or more severe violent crimes) as the threshold. The 78 offenders who passed genotype quality control had committed a total of 1154 murders, manslaughters, attempted homicides or batteries (median 13, range 10–41). There was an overlap between these traits, as the extremely violent offenders were also included in the violent offender group. Homicide offenders (replication cohort) The individuals of the replication cohort were recruited through a forensic mental examination, which is ordered by the Finnish juridical system when a crime is severe, such as homicide, or an exceptionally brutal violent crime. All of these individuals had been convicted for at least one murder or manslaughter. No more detailed information on their criminal history was available. Secondary outcomes in the population-based sample In the Health 2000 sample, the diagnosis of lifetime alcohol use disorders (i.e., alcohol dependence and abuse) was obtained through a structured diagnosis-based evaluation on criteria from the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) for psychiatric disorders. A total of 577 individuals (males n=470, females n=107) in this sample had alcohol use disorder. An early childhood environment of economical difficulties or severe conflicts was retrospectively self-reported by the participants with the questions such as: ‘Did your childhood family have long-lasting financial problems?’ and ‘Were there severe conflicts in your childhood family?’. The possible response options included ‘Yes’, ‘No’ and ‘I don’t know’. Participants reporting ‘I don’t know’ were excluded from the analyses. Altogether, 30% of the sample had reported financial problems or severe conflicts in their family (29% of males, 32% of females). Figure 1 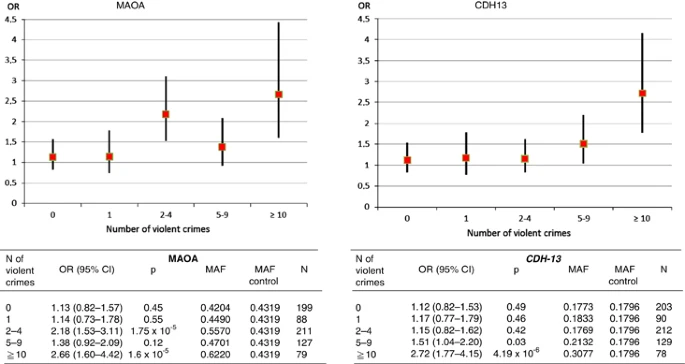 Age- and gender-adjusted odds ratios for low-activity MAOA genotype and CDH13 (rs11649622) as a function of the number of committed violent crimes. The number of individuals, allele frequencies and odds ratios are shown below. The number of control subjects was 1946 for MAOA and 1877 for CDH13 analysis. Genotyping Genotyping procedures are described in Supplementary Information. Results Candidate gene analysis The low-activity MAOA genotype was associated with violent offending in the crime cohort (odds ratio (OR) 1.71, P=2.9 × 10−5; attributable risk 9%, 95% confidence interval (CI) 4–15%). This finding did not differ between males and females, and childhood maltreatment did not modify the risk (OR 1.62; Supplementary Tables 1a and 1b). The association was even stronger among extremely violent offenders (OR 2.66, P=1.6 × 10−4, attributable fraction 16%, 95% CI 8–24%; Figure 1). In the cohort of homicide offenders (N=96), the OR was slightly diluted (1.50, 95% CI 0.82–2.72). No signal was detected from the previously reported HTR2B stop codon SNP (rs798745406) (allele frequency 1.5% among violent offenders vs 0.6% among extremely violent offenders vs 1.6% among controls, P>0.5). |
|
|
|
Post by Admin on Aug 2, 2020 21:08:49 GMT
Genome-wide association analysis We first performed a genome-wide association analysis of violent criminal behavior as two different traits: Violent offending (at least one conviction for violent crime, N=360 for GWAS) and extremely violent offending (10 or more homicides and/or batteries, N=58). In the Supplementary Material, the most significant 50 associations are presented with violent offending and extremely violent offending, respectively (Supplementary Tables 2a and 2b). The genomic inflation factor lambda (λ) for violent offending was 1.0081, and 0.9966 for extremely violent offending, reflecting no significant genomic inflation/deflation in either analysis. The quantile-quantile plot for the observed vs expected results (-2log(P-value)) for the analysis of violent offending resulted in more significant associations than expected by chance, but none of these reached the genome-wide significance threshold of P<5 × 10−8 (Supplementary Figure 2a). In the quantile-quantile plot of the analysis for extremely violent offending, only one data point was observed above the lift off line (Supplementary Figure 2b). The strongest association signals (P<1.0 × 10−5) were observed for the violent offenders for two variants located on chromosome 9q22.1 at the SPIN1 gene (P=3.3−10−6 for rs10123897 and 3.4 × 10−6 for rs11142288), and for the extremely violent offenders for two markers on chromosome 16q23.3 at the CDH13 gene (P=1.7 × 10−6 for rs12919501 and 6.8 × 10−6 for rs4075942), and for single marker at chromosome 9q21.2 at PRUNE2 gene (P=8.5 × 10−6 for rs17787449). These five markers were selected for subsequent genotyping in the extended discovery cohort to increase the sample size and to verify that genotyping with different method had not caused batch effects between the case and the control sample in the discovery (CRIME) cohort. In addition, two additional markers from the CDH13 gene that were among the 25 best genome-wide signals in the extremely violent offenders' group were genotyped in the discovery cohort. Analysis with the regenotyped sample produced concordant genotypes with the original genotyping. Figure 2 shows Manhattan plot in whole genome, and Figure 3 in chromosome 16, with a more detailed view of the SNPs in the region in association with extremely violent offending. The most consistent signal is seen in locus 16q23. The results for six variants within the MAOA gene are shown in Supplementary Table 3. The ORs for the five common variants were 1.1 among the entire violent offender groups, and from 1.4 to 1.7 among the extremely violent offenders (P>0.06). Figure 2 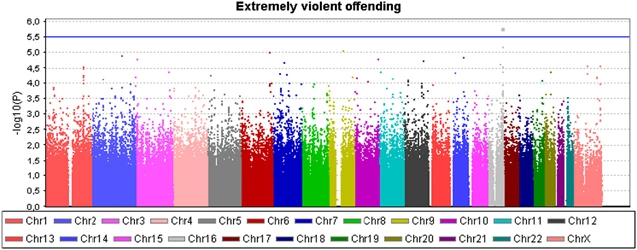 Figure 3 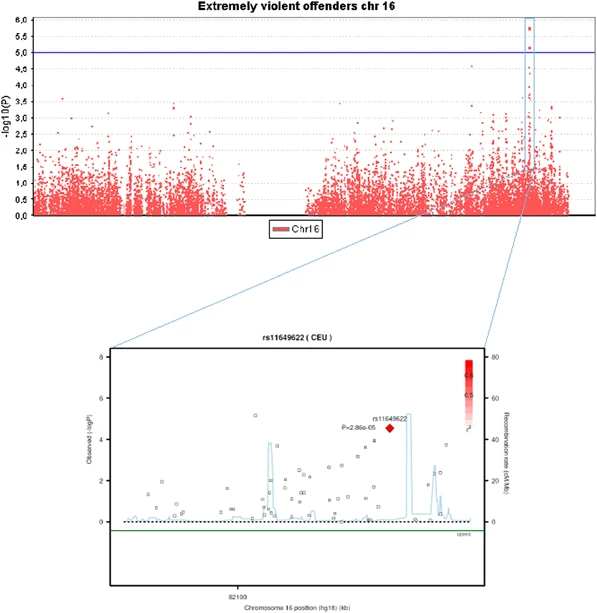 Analysis of single variants for the CRIME cohort and in the replication sample Altogether seven variants were genotyped in the CRIME cohort (N=509) and in 6600 individuals from the Health 2000 sample. Neither of the regenotyping analyses revealed any genome-wide significant signals (P<5 × 10−8), and no suggestive findings (P<1 × 10−5) were observed for an increased risk of violent offending, regardless of whether the individuals had been maltreated or not. The association signal for SPIN1 variants with violent offenders originated from the major alleles and were somewhat diluted with the complete data from the CRIME cohort (P=2.4 × 10−5 for rs10123897 and 1.7 × 10−5 for rs11142288). Analysis of the extremely violent offender data with the complete dataset (n=78 cases) (Supplementary Table 4) shows a suggestive association of P<1.0 × 10−5 and a strengthening of the association signal only for rs11649622, which is located in the intronic region of the CDH13 gene (OR=2.7, P=4.19 × 10−6). Two other CDH13 variants (rs4075942 and rs12919501), located within 100 kb of the most significant variant, had P-values<5 × 10−4. Haplotype analysis of the CDH13 variants provided the strongest evidence for an association of a relatively rare haplotype (f=0.092) of minor alleles A-A from rs11649622 and rs7190768 (P=6.2 × 10−7, OR=4.32; Supplementary Figure 3). The haplotype G-G-A-A covering all the genotyped CDH13 variants (rs12919501, rs4075942, rs11649622 and rs7190768) yielded a P-value of 7.0 × 10−6 (OR=7.21, f=0.030) for extreme violent offending. The variants from CDH13 were genotyped in an independent replication sample of 103 homicide offenders to test for a possible replication association of rs11649622 (as a single variant) and of the variants rs12919501, rs4075942, rs11649622 and rs7190768 (as 2- and 4-nt haplotypes; Table 2). Rs11649622 was associated with homicide offending with P<0.05 (P=0.013; OR 1.73, corresponding to attributable risk of 4%). The haplotypes A-A from rs11649622 and rs7190768, and G-G-A-A from rs12919501, rs4075942, rs11649622 and rs7190768 gave signals of P=0.019 (OR=2.05) and P=0.023 (OR=3.1), respectively, in the replication sample. Thus, the findings for an association of both rs11649622 and CDH13 haplotypes were replicated in the sample of homicide offenders. Post hoc analysis of other single CDH13 variants showed no signal of association (P>0.2) in the replication sample, but there was a signal of association of the haplotype G-G-A from rs11649622, rs12919501 and rs4075942 to homicide offending (P=0.013, OR 2.44). When discovery and replication cohorts were pooled in the meta-analysis, a P-value of 5.3 × 10−7 (OR=2.17) was achieved for rs11649622. Figure 1 shows ORs for rs11649622 among subpopulations of the offenders. Although the risk was not substantially elevated (OR<1.2) among those with 0, 1 or 2–4 violent crimes, it was significantly increased among offenders who had committed 5–9 violent crimes (OR 1.51, 95% CI 1.04, 2.20) or 10 or more crimes (OR 2.72, 95% CI 1.77, 4.15). The overlap between MAOA and CDH13 genotypes among the extremely violent offenders are shown in Supplementary Table 5. The OR for combined homozygosity for low-activity MAOA and rs11649622 minor allele was 13.45 (95% CI 3.86–46.94). |
|












