Post by Admin on Dec 21, 2020 23:33:56 GMT
It was recently shown that the spike protein of SARS-CoV-2 binds to DPP4 (Li et al.
2020) and that DPP4 is a functional receptor for HIV (Li et al. 2020). Moreover, DPP9, a homolog
of DPP4, is associated with severe COVID-19 (Pairo-Castineira et al. 2020). However, one report
suggest that SARS-CoV-2 does not use DPP4 as a receptor (Zhou et al. 2020). Nevertheless, the
current findings suggest that the interaction of SARS-CoV-2 with membrane-bound and secreted
forms of DPP4 deserves further investigation. Inhibitors of DPP4, which can be administered orally
and are used in the treatment of diabetes mellitus, may also deserve attention with respect to possible
effects on viral interactions with the its host cells, as recently pointed out (Scheen 2020, Lim et al.
2021).
It is striking that among eight genetic loci that affect the risk to contract severe COVID19 when
infected with SARS-CoV-2 (Pairo-Castineira et al. 2020), three carry allelic variants derived
from Neandertals. This suggests that the genetic inheritance from Neandertals may have a larger
impact on this disease than would naïvely be expected. However, local adaptation to infectious
diseases often differs among human populations that have been separated by a few tens of thousands
of years (Rees et al. 2020). Neandertals evolved largely independently from modern humans for about
half a million years (Prüfer et al. 2014), even if rare genetic contacts occurred (Kuhlwilm et al. 2016;
Meyer et al. 2016, Posth et al. 2017, Petr et al. 2020). Given the long time of separation, Neandertal
adaptation to infectious diseases may therefore have differed drastically from that of modern humans.
The combination of large effect sizes and small number of Neanderal loci (and
correspondingly smaller number of the multiple tests requiring correction) may allow associations
with infection disease susceptibility to be detected in smaller cohorts than if all variants across the
genome are considered. For the DPP4 locus, we estimate that approximately two times more patients
than currently available in HGI will be needed to achieve a 80% probability to detect the association
between DPP4 and severe COVID-19 with the standard genome-wide significance threshold (p<5e8) (Supplementary Materials).
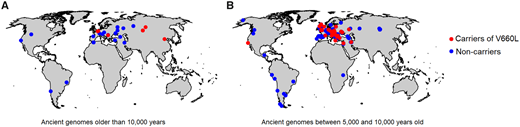
The three Neandertal genomes available to date, which vary in age between ~120,000
years and ~50,000 years and come from Europe and southern Siberia, are all homozygous for the risk
variants on chromosome 2. Furthermore, the late Neandertal genome in Europe, which is most closely
related to the Neandertals that mixed with modern humans, was homozygous also for the risk variants
on chromosome 3 (Prüfer et al. 2017). It is thus likely that the risk variants at both these loci were at
high frequency among late Neandertals. Barring other factors that may affect disease outcome, this
means that, if alive today, a late Neandertal individual would have ~4-16 times higher risk of
becoming critically ill if infected by SARS-CoV-2. This may support speculations that epidemic
diseases could have played a role in the demise of Neandertals.
2020) and that DPP4 is a functional receptor for HIV (Li et al. 2020). Moreover, DPP9, a homolog
of DPP4, is associated with severe COVID-19 (Pairo-Castineira et al. 2020). However, one report
suggest that SARS-CoV-2 does not use DPP4 as a receptor (Zhou et al. 2020). Nevertheless, the
current findings suggest that the interaction of SARS-CoV-2 with membrane-bound and secreted
forms of DPP4 deserves further investigation. Inhibitors of DPP4, which can be administered orally
and are used in the treatment of diabetes mellitus, may also deserve attention with respect to possible
effects on viral interactions with the its host cells, as recently pointed out (Scheen 2020, Lim et al.
2021).
It is striking that among eight genetic loci that affect the risk to contract severe COVID19 when
infected with SARS-CoV-2 (Pairo-Castineira et al. 2020), three carry allelic variants derived
from Neandertals. This suggests that the genetic inheritance from Neandertals may have a larger
impact on this disease than would naïvely be expected. However, local adaptation to infectious
diseases often differs among human populations that have been separated by a few tens of thousands
of years (Rees et al. 2020). Neandertals evolved largely independently from modern humans for about
half a million years (Prüfer et al. 2014), even if rare genetic contacts occurred (Kuhlwilm et al. 2016;
Meyer et al. 2016, Posth et al. 2017, Petr et al. 2020). Given the long time of separation, Neandertal
adaptation to infectious diseases may therefore have differed drastically from that of modern humans.
The combination of large effect sizes and small number of Neanderal loci (and
correspondingly smaller number of the multiple tests requiring correction) may allow associations
with infection disease susceptibility to be detected in smaller cohorts than if all variants across the
genome are considered. For the DPP4 locus, we estimate that approximately two times more patients
than currently available in HGI will be needed to achieve a 80% probability to detect the association
between DPP4 and severe COVID-19 with the standard genome-wide significance threshold (p<5e8) (Supplementary Materials).
The three Neandertal genomes available to date, which vary in age between ~120,000
years and ~50,000 years and come from Europe and southern Siberia, are all homozygous for the risk
variants on chromosome 2. Furthermore, the late Neandertal genome in Europe, which is most closely
related to the Neandertals that mixed with modern humans, was homozygous also for the risk variants
on chromosome 3 (Prüfer et al. 2017). It is thus likely that the risk variants at both these loci were at
high frequency among late Neandertals. Barring other factors that may affect disease outcome, this
means that, if alive today, a late Neandertal individual would have ~4-16 times higher risk of
becoming critically ill if infected by SARS-CoV-2. This may support speculations that epidemic
diseases could have played a role in the demise of Neandertals.