|
|
Post by Admin on Jun 7, 2019 18:00:38 GMT
Description of the specimens The Pyrgos specimen consists of a mandible with partially damaged dentition (c-m3). It belongs to an adult individual as indicated by the fully formed permanent dentition and the closed root apices. The tooth crowns of the right p4-m2 are partially preserved and the dental roots of the right p3-m3 and left c-m3 are largely preserved. The anterior mandibular body is snapped in two, separating both corpora, but the break is clean and the specimen is easily reassembled (Fig 1A, 1C and 1D). Both corpora show slight distortion and some damage, especially on the right side. The mandibular corpus is deep in cross section (tall relative to breadth.) Although the right mandibular corpus is crushed ventrally, a reliable breadth-height ratio is preserved on the left corpus from m2 to m3 (Fig 2, S2 Fig and S1 Table). The mental foramen preserved on the left corpus is positioned below the p4. It is situated ~6.0 mm from the mandibular base and ~22.5 mm from the alveolar margin. The dental arcade is narrow and divergent, with a distance of ~15 mm between the lingual sides of the p3 cervicesand ~26 mm at the m3s (Fig 3A and S2 Table).  Fig 2. Robusticity and dimensions of the mandibular corpus in G. freybergi and O. macedoniensis. a, Mandibular robusticity index (RI = corpus breadth/height) in different tooth positions of G. freybergi compared to female and male O. macedoniensis (RPL-54, NKT-21, RPL-90, RPL-80, RPL-56, RPL-75, RPL-89, RPL-94; [47, 48] and this study). b, Corpus breadth and c, Corpus height in different tooth positions of G. freybergi, O. macedoniensis (RPL-54, NKT-21, RPL-90, RPL-80, RPL-56, RPL-75, RPL-89, RPL-94; [47, 48] and this study) and Au. afarensis [52]. In G. freybergi, the mandibular corpus is laterally crushed and is close to the real breadth only posterior to the left m2. Minimum estimations are indicated with dashed line.  Fig 3. Morphometry of the mandibular corpus and symphysis in G. freybergi and O. macedoniensis. a, Bivariate plot of the mandibular tooth row of G. freybergi (black) and O. macedoniensis (RPl-54; grey) illustrating the differences in arcade width. The lingual distances between the left and right tooth row are plotted for each tooth position, if preserved. Measurement was done at the lingual sides of the dental cervices. The vertical axis shows the measuring position along the tooth row given as distance from m3. b, Top: Sagittal sections through mandibular symphyses of O. macedoniensis (RPl-56, RPl 75, RPl-54 [53]). Bottom: Sagittal section through the preserved veneer of the mandibular symphysis of G. freybergi (black) aligned to the symphysal sagittal section of O. macedoniensis [53] (RPl-54, grey; same scale). AP: Alveolar plane; TP: plane of transverse tori; SP: sublingual plane. The inclination of TP and SP, and the angle between both planes is given for G. freybergi. The symphysis provides only limited information as it is mostly missing save a thin veneer (2–3 mm) of a portion of the lingual cortical bone surface (Fig 3B). The CT scans show that the anterior cortical and trabecular bone are missing and confirm that some cortical bone of the internal (lingual) surface is preserved. Hence, the lower part of the sublingual plane, the superior transverse torus (t.t.sup.) and the inferior transverse torus (t.t.inf.) are preserved (Fig 3B). The genioglossal fossa between both tori is shallow but clearly visible. The horizontal position of the t.t.sup. is at the level of the mid-p4, and the t.t.inf. is at the level between p4 and m1. The constructed bitangent of the t.t.sup. and t.t.inf. (plane of transverse tori; TP) forms an angle of 56° with the alveolar plane. The sublingual plane is oriented at an inclination of about 37°. The symphysal height and depth are not measurable.  Fig 4. Dental crown dimensions of Late Miocene hominids. a, m2 crown dimensions of G. freybergi, O. macedoniensis, O. turkae, N. nakayamai and A. meteai. Comparative data: [41, 47–50, 55]. b, P4 crown dimensions of cf. Graecopithecus sp., O. macedoniensis and O. turkae. Comparative data: [39, 41, 48, 56]. The partially preserved crowns of right p4 (its mesiobuccal face is missing), m1 and m2 show extreme occlusal and interstitial wear (Fig 1C). The p4 retains only a thin layer of occlusal enamel. Dentine is exposed on its buccodistal half and the metaconid (wear stage 5, after [67]). Although the occlusal surface is largely flattened, a mesio-distal step is clearly visible between the mesial cusps and the talonid. The occlusal enamel of the m1 and m2 is almost completely worn away, exposing large parts of the dentine. In m1 the conids are entirely worn away and only the outer rim of enamel remains (wear stage 7). In m2 (wear stage 5–6) the abrasion is focused on the buccal conids, where a deep hollow reaches the pulp chamber. The entoconid and metaconid are still visible, but expose their dentine horns. Due to the interstitial wear the mesial face in m1 is S-shaped and in m2 concave (Fig 1C). The distal half of m1 is obliterated with the interstitial wear reaching deep into the dentine. Martin & Andrews [45] calculated a crown length reduction of 32% for this tooth. This is consistent with the estimated loss of 30% in m1 tooth area, when we apply the tooth size prediction after Evans et al. [54]. Reliable crown measurements can only be taken from m2 (BL = 13.2 mm, MD≈14.2 mm; Fig 4) and p4 (MD = 9.1 mm). Based on the cervical root areas the tooth size is estimated to increase from m1 to m3 [45]. The m2 is often referred to as being slightly broader than the mandibular corpus at this level [38, 44–46], which is seen as a unique character of G. freybergi. However, this is partially an artefact of crushing, as the μCT-section reveals (S2 Fig). The better-preserved left corpus shows a breadth similar to that of m2, which is nevertheless unique among hominoids. Hence, the posterior dentition still shows a clear evidence of megadontia relative to corpus dimensions, but perhaps less dramatically than previously thought. The teeth are thickly enamelled, with a lingual radial enamel thickness of 1.40 mm for m2 and 1.50 mm for p4 (Fig 5 and S5 Table). The m1 radial enamel thickness is not measurable. The pulp chambers of the molars (right m1 and m2; S1C Fig) are vertically narrow. Their upper surface is flat as their pulp horns are inconspicuous or lacking. The CT-scans reveal an accumulation of dentine in large parts of the pulp chamber and pulp horns. Dentine layers of less density may trace the original pulp chamber. Thus, an accretion of secondary dentine can be assumed, particularly on the roof and the horns of the pulp chambers.  Fig 5. Radial enamel thickness in m2 of extant and extinct hominoids including G. freybergi. The lingual radial enamel thickness (l) in G. freybergi is measured on μCT slices at the lingual side of the metaconid, following [58]. Comparative data: [41, 58–60]. Horizontal line = mean; vertical line = range. The maximal root lengths (longest root of a tooth, measured on 3D) of the molars are (left/right) m1 >13.5/ = 14.5 mm; m2 >16.9/ = 17.6 mm; m3 = 15.6/16.9 mm. The left canine root (>16.1 mm) is partially preserved, but its upper mesial part is missing. However, it is possible to estimate its maximal length to the cervical plane (c ≈25.5 mm; S3 Fig). RIM 438/387 –the left P4 from Azmaka [39] has an intensively worn crown and three well preserved roots (Fig 1B). The crown is mesio-distally narrow with rounded rectangular occlusal outline (MD = 8.2 mm; BL = 12.3 mm). The enamel is thick with a buccal radial thickness of k = 1.55 mm. The occlusal wear facet is mesio-labially inclined and exposes large parts of the lingual dentine (wear stage 4; after [67]), but only the tip of the buccal dentine horn (wear stage 2). The distal crown surface shows a distinct interstitial wear facet. The P4 has a maximal root length of 12.0 mm; its roots are mesio-distally compressed. The buccal roots are close to each other and are fused in the upper 3 mm. Each radical features a separate pulp canal (S1A Fig). |
|
|
|
Post by Admin on Jun 8, 2019 18:52:27 GMT
Comparison and taxonomic validity G. freybergi is only known from one mandible and possibly the tooth from Azmaka (Fig 1A and 1B). This compares with a relatively large number of Ouranopithecus specimens. Ouranopithecus has been synonymised with Graecopithecus by some [45]. Others emphasize the dentognathic differences between both taxa, but regard the Pyrgos specimen as largely uninformative due to its poor surface preservation and vague dating [44]. The new data provided here support previous conclusions that Ouranopithecus and Graecopithecus differ in significant numbers of characters more than adequate to recognize two different taxa with probable generic differences [41]. Beside shared characters between G. freybergi and O. macedoniensis (thick enamel [44, 68, 69], m2 crown dimension, symphyseal shape; Figs 3 and 4), both taxa differ in the dental arch, which is shorter and narrower in G. freybergi (Fig 3A). The width (BL) and length (MD) of the m2 crown is within the range of female O. macedoniensis (Fig 4 and S3 Table), but it is broader relative to the mandibular robusticity. The BL width of m2 approximates the breadth of the mandibular corpus at this position. Hence, the mandible of G. freybergi is very gracile compared to O. macedoniensis and other Miocene and Pliocene hominids (Fig 2 and S1 Table), as already suggested by von Koenigswald [38] and Martin & Andrews [45]. Generally, the mandibular corpus breadth in hominids show only minor sex differences, but is of taxonomic significance [70–72]. The breadth of female and male O. macedonienis mandibles are closer to one another than either is to G. freybergi (Fig 2B). Thus, the considerable lower breadth in G. freybergi strongly suggests a taxonomic difference. In contrast, the mandibular robusticity is significant for sex discrimination in hominids [35, 70, 71]. Male O. macedoniensis are less robust (taller relative to breadth) than females. The mandibular height of G. freybergi overlaps with the height of female O. macedoniensis, but its robusticity is in the lower range of the gracile males (Fig 2A). Assuming a similar pattern of sexual dimorphism with robust mandibles in females and gracile mandibles in males, the very gracile mandible of G. freybergi relative to its m2 size and compared to O. macedoniensis and other Miocene and Pliocene hominids (S1 Table), suggests that the Graecopithecus type mandible may belong to a male individual. G. freybergi and O. macedoniensis differ in the number of their dental roots and/or pulp canals (Table 1) showing a reduced configuration in G. freybergi. Further, the buccal fusion of the p4 roots differs from the separated roots in O. macedoniensis and other Late Miocene hominids (e.g. O. turkae; see figure 2 in [41]), but approximates the root form recently described in australopithecine specimens from Woranso-Mille, in Au. africanus and in P. robustus [25, 36]. Much variability is known for the root number and morphology within and among australopithecine species, from a Tomes’ root to a three-rooted morphology (e.g. [26, 36]). However, within the fossil record the p4 root fusion is a feature that appears exclusively in hominins. 12% of P. robustus (n = 2) and ~17% of Au. africanus (n = 3) have either a fused p4 root or a single root [36]. There is no example of any root fusion (partial or complete) in the p4 of non-hominin fossil apes, and there are only very rare occurrences in Pan. In the large tooth samples of extant Pan observed in several studies, the hominin condition is present in less than 2–5% [63, 73, 74]. Further, the root configuration in p4 is less variable than in other lower and upper premolars of Pan [63]. The inter-genus variability among extant great apes is low, but large between great apes and humans.  Table 1. Root and pulp canal configuration in c-m3 of G. freybergi (holotype, this study) and O. macedoniensis [32]. Similar to O. macedoniensis, the root lengths are rather short compared to extant great apes [32]. In G. freybergi, this particularly concerns the canine and m1. The absolute canine root length (Fig 6 and S6 Table) is below S. tchadensis and in the range of Au. anamensis, Ar. ramidus and female P. troglodytes. Given that G. freybergi may be a male individual, the short canine root may indicate canine reduction. However, this observation needs further confirmation by more canine root length data. The m1 root length is in the range of P. troglodytes and H. sapiens, but considerably below Gorilla and S. tchadensis. While in extant great apes and S. tchadensis the root length of m1 is similar to m2, G. freybergi shows an m1 root that is considerably shorter than those of m2 and m3.  Fig 6. Absolute root lengths of the lower dentition in extant and extinct hominids. The root lengths of extant great apes and humans (min/max/mean/S.D.) are plotted in colour for both sexes (left: males, right: female). The root lengths of fossil taxa (min/max/mean; grey shaded) are sex-pooled. The sample size (n) of fossil taxa is indicated within the graph. For the type of G. freybergi the root lengths of both hemimandibles are plotted, if preserved. Dashed lines are estimations (see also S3 Fig). Comparative literature data: extant great apes and humans [64]; Au. afarensis and Au. anamensis [65]; Ar. ramidus [31]; S. tchadensis [28]. Detailed data in S6 Table. In G. freybergi, the radial enamel thickness of the m2 is considerably greater than in extant great apes and Griphopithecus alpani (Fig 5 and S5 Table). With l = 1.40 mm it is close to Griphopithecus darwini (1.23 mm) and within the mid-range of more thickly enamelled hominins (e.g. Homo sapiens l = 1.41 mm). Ouranopithecus turkae shows a considerably higher value of l = 2.08 mm. O. macedoniensis has also a very thick molar enamel [68, 69]. The literature on O. macedoniensis is not directly comparable to our measurements. However, its relative and absolute molar enamel thickness is reported to exceed that of extant great apes and other Miocene hominids [69]. The P4 from Azmaka, Bulgaria is nearly contemporaneous (~65kyr older) with G. freybergi from Pyrgos [40]. Previously, the P4 had been referred to cf. Ouranopithecus sp. or aff. G. freybergi [39]. This study shows that some morphological aspects are indeed shared with G. freybergi. The P4 is thickly enamelled, showing the same radial enamel thickness (k = 1.55 mm) as the p4 from Pyrgos (l = 1.50mm). While the size of the Azmaka P4-crown (BL = 12.3 mm; MD = 8.2 mm; Fig 4B) is similar to female O. macedoniensis (BL = 12.5–13.3 mm; MD = 7.25–9.0 mm), its roots are less robust and more parallel, as in the roots of G. freybergi. The P4 roots of the female and the larger sized roots of male O. macedoniensis are more separated and diverge towards the apex (Fig 7). Hence, both individuals from Azmaka and Pyrgos show the same evolutionary trend in upper and lower teeth respectively. Accordingly, we assign the Azmaka specimen to cf. Graecopithecus sp.  Fig 7. Root morphology in P4 of cf. Graecopithecus sp. and O. macedoniensis. Top row: Left P4 of cf. Graecopithecus sp. from Azmaka (Bulgaria) in distal, buccal and apical view. Below: Female and male specimens of O. macedoniensis from Ravin de la Pluie (Greece). The pictures of the right P4 (RPl-101) are mirrored for a better comparison. (Photos of O. macedoniensis teeth made with courtesy of G. D. Koufos, Aristotle University of Thessaloniki). Differential diagnosis G. freybergi differs from extant great apes (Pan, Gorilla, Pongo) in its thickly-enamelled teeth (Fig 5). It differs from the similar sized P. troglodytes in its absolutely longer dental roots of m2 and m3, but shows comparable c to m1 root lengths (Fig 6). G. freybergi differs from most hominids (e.g. Sivapithecus, Ouranopithecus, australopiths, early Homo) in its gracile mandibular corpus (Fig 2). Its corpus height is within the lower range of female O. macedoniensis, but its breadth is lower. It can be further distinguished from O. macedoniensis by its narrow dental arc (Fig 3). G. freybergi differs from O. macedoniensis in its root configuration, having two-rooted lower premolars including a partially fused p4-root and a reduced number of pulp canals (note the considerations on intra/inter species variation below). It differs from Ouranopithecus turkae in having absolutely and relatively thinner enamel and a fused p4-root. The m2 crown size (MD = 14.2mm; BL = 13.2mm) is intermediate between female and male O. turkae. Emended diagnosis G. freybergi is a hominid in the size range of female chimpanzees based on dentognathic size. The mandibular dental arch is anteriorly narrow (lingual distance between p3s ≈ 15mm) and diverges slightly posteriorly (lingual distance between m3s ≈ 26mm). The symphysis shows a weak upper and lower transvers torus and a sublingual plane at about 37° relative to the alveolar plane. The mandibular corpus is narrow and deep, which results in a low robusticity index (RI = 0.53 at m2). The posterior dentition is megadont relative to corpus size, with a broad m2 that matches the breadth of the mandibular corpus in this position. Tooth size is estimated to increase from m1 to m3, based mainly on the cervical root area. The enamel is thick (Fig 5 and S5 Table). The dental roots of the tooth row (c to m3) are short (c ≈25.5 mm; p3 ≈16.5 mm; p4 ≈15.9 mm; m1 ≈13.6/ = 14.5 mm; m2 ≈18.0/ = 17.6 mm; m3 = 15.6/16.9 mm; maximum length of left and/or right dentition, derived from μCT based 3D reconstructions, see S3 Fig and S6 Table). The premolars and m3 are two-rooted. The p4 shows a fusion of the mesial and distal root in the upper buccal part. The m1 is three-rooted; the m2 shows three (left) or two (right) roots. Both, m1 and m2 show bifurcated apices in their mesial roots. The molars have low pulp chambers with blunt pulp horns. The number of pulp canals in the postcanine teeth is low (Table 1). |
|
|
|
Post by Admin on Jun 8, 2019 20:00:51 GMT
 Phylogenetic position of Graecopithecus The investigation of the internal structures of the Pyrgos mandible reveals characters of the roots of the p4 that are derived compared to other Miocene apes and extant great apes. In contrast to the Ponginae, Graecopithecus shares derived characters with African apes (ventrally shallow roots, buccolingually broad molar roots; [32, 75]). Therefore, we consider four principle alternative interpretations of its phylogenetic position: Graecopithecus is a stem-hominine (last common ancestor of African apes and Homo), a gorillin, a panin, or a hominin. Basal hominids like Proconsul have two or three clearly diverging roots and four pulp canals (1-22M+12D) in the p4 [28]. The prevailing root configuration in extant great apes is two roots and two to three pulp canals [73], which is the condition seen in G. freybergi (11M+12D). However, the mesial and the distal roots of G. freybergi are partially fused at about 47% of maximal root length (Fig 8), a character which is extremely rarely observed in extant great apes (2–4%; [73]). This fusion may represent an early stage of a Tomes’ root, a character that is considered diagnostic for the hominin clade [26, 27]. Thus far, a buccal root fusion similar to G. freybergi is reported from australopithecines [25, 36]. The configuration of the p4 root and the pulp canal in G. freybergi is intermediate between the narrow p4 roots in S. tchadensis [28] (Fig 8) and the Tomes’ root in Ar. kadabba [76]. The derived state of G. freybergi with respect to O. macedonensis is further supported by root and pulp canal reductions in other tooth positions (Table 1). The hominin record shows different levels of p4 root fusion, although separated roots are common as well. However, p4 root fusion never occurs in Miocene non-hominins, suggesting that this feature in Graecopithecus is a hominin synapomorphy. Accordingly, the most parsimonious interpretation of the phylogenetic position of Graecopithecus is that it is a hominin, although we acknowledge that the known sample of fossil hominin root configurations is too small for definitive conclusions.  Fig 8. Root morphology of the lower fourth premolar (p4) in Graecopithecus and Sahelanthropus. a, Cervical μCT-section through the right mandibles of S. tchadensis (left; [29]) and G. freybergi (right) with drawings of their p4 cross-sections at the level just below the cervix (for G. freybergi 2.5 mm below p4 cervix). b, Root configuration in p4 of G. freybergi. The apical parts of the right p4 roots are missing, but an approximate reconstruction was done by aligning the mirrored roots of the left p4 (in transparent blue). The left p4 is broken just below the level of bifurcation. LB = height of lingual bifurcation, BB = height of buccal bifurcation (both preserved on the right p4). Scale bar, 10 mm. A feature supporting this interpretation is the observation of canine root reduction. With an estimate canine root length of ~25.5 mm (Fig 6), the probably male specimen of G. freybergi is in the range of female P. troglodytes (24.1 ±2.7 mm [64]) and below female G. gorilla (29.4 ±2.2 mm). It is in the range of Au. anamensis (20.3–31.8 mm [65]) and Ar. ramidus (25.0–31.4 mm [31]). Further, it is shorter than the lower canine root of S. tchadensis (27.97mm [28]) and above Au. afarensis (21.0–24.3 mm [65]) and H. sapiens (16.5±2.1mm [64]). 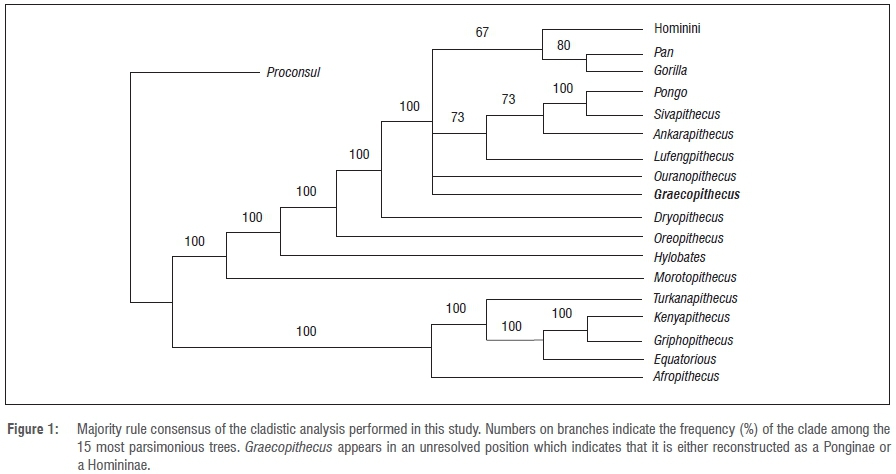 In earlier studies, a relationship of European hominids to the African hominins is proposed [77, 78]. Taken at face value, the derived characters of Graecopithecus (p4 root morphology and possibly canine root length) may indicate the presence of a hominin in the Balkans at 7.2 Ma. In many publications, de Bonis, Koufos and colleagues have proposed that Ouranopithecus, from northern Greece and more than 1.5 million years older, is a hominin [47, 79, 80]. Other researchers have interpreted the similarities between Ouranopithecus and australopithecines as homoplasies [81]. It is possible that the similarities between Graecopithecus and Ardipithecus and some australopithecines are also homoplasies. However, as stated before the premolar root number is less functionally constrained than megadonty and enamel thickness, and thus, potentially more useful for phylogeny reconstruction [19, 20]. Graecopithecus has reduced root morphology yet heavy mastication and megadontia, suggesting a de-coupling of root and molar function. In contrast, larger roots, large teeth and thicker enamel together contribute to a functional complex shared with australopithecines, which is evoked as the mechanism accounting for the homoplastic appearance of hard object feeding adaptations in Ouranopithecus and australopithecines [81]. Therefore, we submit that the dental root attributes of Graecopithecus suggest hominin affinities, such that its hominin status cannot be excluded. If this status is confirmed by additional fossil evidence, Graecopithecus would be the oldest known hominin and the oldest known crown hominine, as the evidence for the gorillin status of Chororapithecus is much weaker than the hominin status of Graecopithecus [8]. More fossils are needed but at this point it seems likely that the Eastern Mediterranean needs to be considered as just as likely a place of hominine diversification and hominin origins as tropical Africa. Fuss J, Spassov N, Begun DR, Böhme M (2017) Potential hominin affinities of Graecopithecus from the Late Miocene of Europe. PLoS ONE 12(5): e0177127. |
|
|
|
Post by Admin on Oct 29, 2020 19:36:44 GMT
Research articles Dissecting human North African gene-flow into its western coastal surroundings Lara R. Arauna, Garrett Hellenthal and David Comas Published:01 May 2019 doi.org/10.1098/rspb.2019.0471Abstract North African history and populations have exerted a pivotal influence on surrounding geographical regions, although scant genetic studies have addressed this issue. Our aim is to understand human historical migrations in the coastal surroundings of North Africa. We built a refined genome-wide dataset of North African populations to unearth the fine-scale genetic structure of the region, using haplotype information. The results suggest that the gene-flow from North Africa into the European Mediterranean coast (Tuscany and the Iberian Peninsula) arrived mainly from the Mediterranean coast of North Africa. In Tuscany, this North African admixture date estimate suggests the movement of peoples during the fall of the Roman Empire around the fourth century. In the Iberian Peninsula, the North African component probably reflects the impact of the Arab expansion since the seventh century and the subsequent expansion of the Christian Kingdoms. By contrast, the North African component in the Canary Islands has a source genetically related to present-day people from the Atlantic North African coast. We also find sub-Saharan gene-flow from the Senegambia region in the Canary Islands. Specifically, we detect a complex signal of admixture involving Atlantic, Senegambian and European sources intermixing around the fifteenth century, soon after the Castilian conquest. Our results highlight the differential genetic influence of North Africa into the surrounding coast and show that specific historical events have not only had a socio-cultural impact but additionally modified the gene pool of the populations. 1. Introduction North Africa is a genetically diverse region from a human population perspective. North African populations show a complex and heterogeneous genetic structure that has been described as an amalgam of at least four different ancestral components: Middle Eastern, sub-Saharan African, European and autochthonous North African [1]. Most of the genetic studies about North Africa have focused on the inner relationships among populations, or the gene-flow from nearby populations [1,2]. However, there are scant studies that have focused on North African gene-flow into neighbouring regions [3]. It is well known that the surrounding coast has been historically influenced by North African peoples [4,5]; however, the demographic impact of those contacts has not been properly addressed. Our aim is to assess the North African demographic and genetic influence in nearby regions outside the African continent by assessing the gene-flow in three geographical neighbouring regions with documented contacts with North Africa: the Canary Islands, the Iberian Peninsula and Tuscany. The Canary Islands, located in the Atlantic coast of North Africa, have been inhabited since approximately 1000 BCE [5,6]. The islands were known by the Phoenicians, Greeks and Romans; however, it is thought that there was no contact with the autochthonous settlers of the islands since the fourth century until the Castilian conquest in the fifteenth century [7]. By the time of this European conquest of the Islands, the aboriginal population size has been estimated around 100 000 individuals [8]. A northwest African origin of the first settlers of the islands is consistent with patterns of uniparental and classical genetic markers in modern and ancient samples [9,10]. In particular, the presence of haplogroups in the Canary Islands that are only found in individuals of North African descent, such as mitochondrial (mtDNA) haplogroup U6 [11] and Y-chromosome haplogroup M81 [12], among some others considered founder lineages, support the North African origin of the islanders. The frequencies of these haplogroups in the extant population of the Canary Islands show a clear sexual bias: the percentage of the maternal North African component estimated through the analysis of mtDNA lineages is high, between 42 and 74% [10]; while the paternal component analysed through the study of Y-chromosome lineages is lower, between 5 and 16% [9]. Additionally, Botigué et al. [3], analysing genome-wide data, showed a higher identity by descent sharing between individuals from the Canary Islands and North Africa compared to individuals from continental Europe, suggesting a higher gene-flow from the African continent to the Islands. Finally, genome-wide analysis with ancient DNA from the Canary Islands has corroborated the North African origin of the autochthonous component and its presence in current Canary Islanders [13]. However, the exact dates of admixture from Europe and the precise geographical origin of the North African component in the Islands have not been addressed. The most well-documented contact between North Africa and the Iberian Peninsula is the Arab expansion, which crossed the Mediterranean and arrived in Gibraltar in the eighth century. However, genetic studies based on uniparental markers, together with archaeological and anthropological evidence, have suggested previous contacts across the Gibraltar Strait that date back to prehistoric times [4,5,11,14,15]. Recently, ancient DNA studies have supported prehistoric migrations from North Africa into the Iberian Peninsula since around 4000 ya (years ago) [16–18]. Moreover, mtDNA, Y-chromosome and short tandem repeat studies [4,19–22], as well as genome-wide and ancient DNA analyses [3,17,23], have also shown gene-flow in historical times. Moorjani et al. [23] dated African gene-flow into southern Europe around 55 generations ago, with the highest proportions in Iberia: 3.2 ± 0.3% in Portugal, and 2.4 ± 0.3% in Spain, which was related to a demographic impact either in Roman or Arab periods. Botigué et al. [3] showed that the inclusion of North African populations in their analyses increased those estimated percentages of gene-flow, suggesting a higher North African gene-flow in Iberia, and that the sub-Saharan gene-flow detected entered with the North African wave, challenging the interpretation of a direct sub-Saharan influence in southern Europe. Additionally, the North African gene-flow in the Iberian Peninsula was dated to 6–10 generations ago, although previous gene-flow was not discarded. In a large study of human populations admixture, Hellenthal et al. [24] described a complex scenario with continuous gene-flow during the past 2000 years in Iberia with North and sub-Saharan Africans. In sum, although all studies agree on the genetic influence of North Africa in Iberia, there is no clear consensus in the pattern of gene-flow and the estimated dates of the North African admixture. The presence of the Etruscans, in what is nowadays referred to as the Tuscany territory in the Italian Peninsula, has been largely documented. However, the genetic footprint of the Etruscans in current populations has been only claimed in some isolated populations, but not in the Tuscan general population [25]. Moreover, although a Middle Eastern or Anatolian origin has been hypothesized for the Etruscans [26], recent studies analysing mtDNA have rejected an origin outside Italy [25,27]. Recently, an exhaustive genome-wide study of the Italian population [28] has dated different admixture events in Italy coming from different sources, including old events dated around 3000 ya that involved Caucasus, Middle Eastern and central Italian populations; whereas other more recent admixture processes involved gene-flow from north-central Europe around the collapse of the Roman Empire, a period which has been associated with extensive human movements. This continuous gene-flow in multiple directions at different times has yielded a complex genetic structure in the Italian Peninsula shown in both uniparental and genome-wide analyses [28,29], and traces of North African influences have also been detected, although the amount and timing of such contributions to Italy have not been assessed [28,30]. Our aim is to assess the impact of gene-flow from North Africa to surrounding populations for which there is documented evidence of contact between the populations, in particular the Canary Islands, Iberia and Tuscany. Previous North African–European gene-flow analyses [3,23] were limited by the scant geographical distribution of North African samples available. In order to overcome these issues, we use the largest genome-wide dataset of North African samples available, including different Berber groups that have not been included in previous studies of North African gene-flow, which allows us to describe detailed and complete genetic scenarios for North African admixture into the surrounding areas. The application of haplotype-based methods to a large dataset of samples and autosomal markers might refine our knowledge on the (i) estimated dates of the admixture events; (ii) the specific geographical sources of the gene-flow; and (iii) the quantification of the amount of gene-flow in the three targeted populations. |
|
|
|
Post by Admin on Oct 30, 2020 0:42:44 GMT
Results We have compiled a dataset of more than 1200 samples that includes a large and diverse dataset of North African populations to study the influence of North African gene-flow in neighbouring populations. Principal components analysis (PCA) of all populations in the dataset differentiates sub-Saharan and European populations along the first PC (PC1) (figure 1). The North African samples are widely spread along the first PC reflecting high heterogeneity, in accordance with the previously described differential admixture of the subpopulations [1,2]. PC2 further differentiates North African samples and highlights the genetic diversity within North Africa. On the first two PCs, the Canary Islands samples are placed close to the Iberian samples but shifted towards the Middle East and North Africa. When focusing on the European samples, three largely non-overlapping clusters can be observed: the Finns; northern and western Europeans (Great Britain and Utah residents with northern and western European ancestry (CEU)); and southern Europeans (Tuscany, Iberia, Basque Country and also the Canary Islands).  Figure 1. Principal component analysis (PCA) of the samples included in the present study. See material and methods section for details. We used haplotype-based methods to dissect the genetic structure of the studied populations and understand their genetic relationships. We performed fineStructure analyses (figure 2) and identified three major splits separating our data: North Africa, Europe and sub-Saharan Africa. Within these major geographical clusters, several subclusters can be identified that suggest a finer resolution of genetic structure. For example, within the European cluster, six sub-clusters are found that correlate with geography: Iberian Peninsula, Tuscany, Basque, Canary Islands, northwest Europe and Finland (electronic supplementary material, figure S1). Thirteen Syrian samples clustered together with the Canary Islands populations and were removed from further analyses. Similarly, within sub-Saharan African samples we find four sub-clusters that correspond closely with sampling locations: Luhya (from Kenya), sub-Saharan Atlantic (GWD and MSL), Guinean Gulf (YRI and ESN) and North Africa_sub-Saharan_ancestry, which is composed of North African samples with substantial sub-Saharan admixture (as previously described in [2]). By contrast, sub-clusters within North Africa do not show as precise a correlation with geography, with several sub-clusters containing individuals that span broad geographical areas: east, west, central, Atlantic, Mediterranean, Tunisia Chenini and Tunisa Sened (the last two have been already described as drifted populations that show high levels of relatedness [1,2] (figure 2 and electronic supplementary material, figure S2). Finally, a dissection of the Iberian Peninsula sub-cluster shows four minor clusters: NorthWest_Iberian, South_Iberian and two clusters without clear geographical structure (Iberian_Peninsula1 and Iberian_Peninsula2; electronic supplementary material, figure S3). One Iberian individual was an outlier (did not cluster), and therefore this individual was not included in further analyses.  Figure 2. fineStructure clustering shown as a dendrogram and its correspondence in a map. The filled rectangles are the North African samples, and the proportion of individuals from each of the clusters in each geographical sampled population is shown in pie-charts. Clusters containing European and sub-Saharan African individuals are denoted by non-filled rectangles coloured blue and yellow, respectively, and are labelled primarily according to geography. |
|








