|
|
Post by Admin on Jan 18, 2021 20:27:20 GMT
Results and discussion We have assembled data on 238 population samples with allele or genotype frequencies for at least one of the four commonly studied variants. Most of those studies have data on two or more of the SNPs (Table S1). 105 population samples have data for all four of those SNPs at OCA2: three amino acid substitution SNPs at OCA2, rs1800414, rs74653330, and rs1800407 and the OCA2 enhancer SNP, rs12913832, in an intron of HERC2. The population samples with OCA2 data are listed in Supplemental Table S1. Individual SNP frequencies The population specific allele frequencies of the four functional SNPs noted in Table 1 are given in Supplemental Table S1 and presented as density plots in Figs. 1, 2, 3 and 4; a different graphic representation indicating the frequency data for each specific population sample is given in Supplemental Figs. S1 through S4. All of the functional SNPs have data for many population samples. Each of the Supplemental figures includes all of the population samples with data for any of the SNPs; blanks represent missing data for a given population sample. Each bar in the Supplemental figures represents the data from a single population study involving that SNP; there are several instances of multiple independent samples for the same ethnic/geographic group. Figure 1 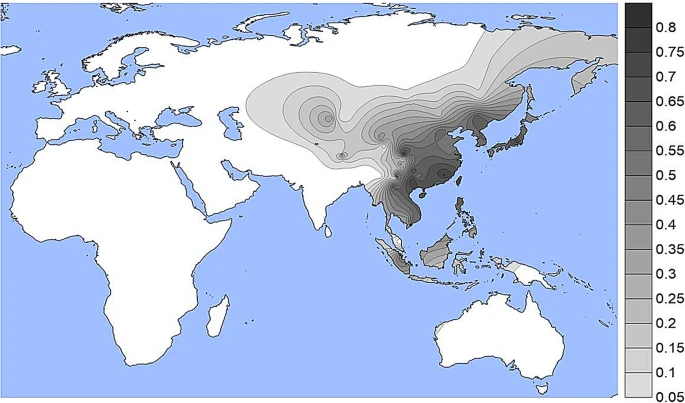 A density plot of the frequencies of the derived allele at rs1800414. The underlying data for Figs. 1, 2, 3 and 4 are in Table S1. Alternative graphic representation with the frequencies of each population sample is in Fig. S1. See text. The derived allele at rs1800414 is largely restricted to but common in many East Asian populations (Figs. 1 and S1). This SNP has been studied in many populations that have not been studied for various of the other three SNPs. This variant reaches frequencies over 50% in most of East and Southeast Asia. It has lower frequencies of 5% to 15% in the Pacific populations and in Central and Northern Asia as well as Tibet and other parts of Southwestern China. The derived allele at the missense SNP, rs74653330 (Ala481Thr) (Figs. 2 and S2) has been studied less comprehensively than rs1800414 but occurs widely in Northern Eurasia and is especially common in Eastern Siberian and Mongolian populations The report of a frequency of 52% in the Oroqen (sampled in northern China near the Russian border) is an outlier in terms of frequency but not geography: it was omitted from Fig. 2 but not Fig. S2. Off the scale of Fig. 2 (frequencies < 4%) the derived allele occurs rarely in most of Europe, in some Southwest Asian populations (Turkish, Iranians), in South Asia (Hazara), and in China (Tibetans). In northern Europe it occurs at low frequencies (1% to 3%) in some populations (Chuvash, Vologda Russians) and reaches 5% to 7% in Finnish samples. Given that the derived allele at rs74653330 is hypomorphic, it is a clear candidate for studies of selection favoring the allele in the northern populations. Figure 2 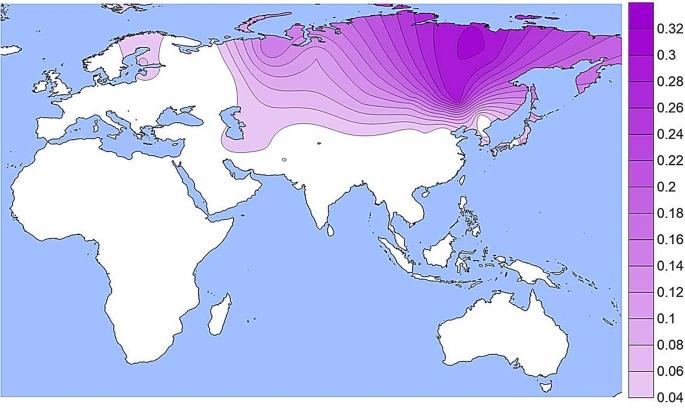 A density plot of the frequencies of the derived allele at rs74653330. The scale has been adjusted to minimize visual extrapolation to very rare occurrences. An outlier frequency of 0.52 in a small Orogen sample was omitted from the density plot and the omission resulted is a slight shift of the highest frequency region to the West. See Figure S2, caption for Fig. 1, and text. |
|
|
|
Post by Admin on Jan 19, 2021 3:50:50 GMT
The derived allele at rs1800407 (Figs. 3 and S3) occurs at low frequencies in most populations in North Africa, Europe, South Asia, and in some populations in East Asia but mostly off the scale in Fig. 3 which is driven primarily by a few values greater than 10% frequency. For example, in 18 Spanish Basques the frequency is 21% while in 14 Orcadians, the frequency is 14%. Figure 3 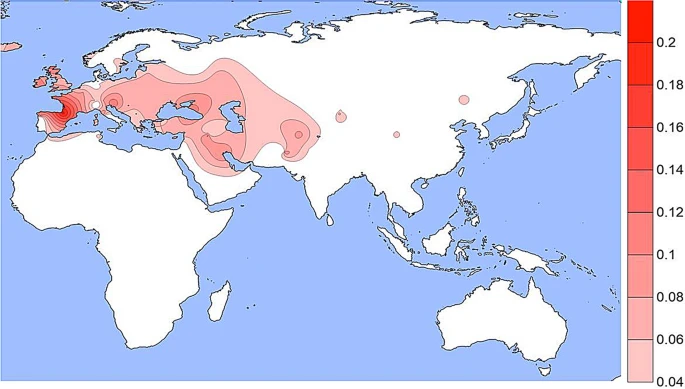 A density plot of the frequencies of the derived allele at rs1800407. The scale has been adjusted to minimize visual extrapolation to very rare occurrences. See Fig. S3, caption for Fig. 1, and text. The rs12913832 SNP (Figs. 4 and S4) is the enhancer polymorphism and has the largest number of population samples with data since most studies of other pigmentation SNPs have also included rs12913832. This variant is well known for high frequencies in Northern Europe (70% to 95%) as seen in Figs. 4 and S4. It is found at more moderate frequencies in populations from Southern Europe, Southwest Asia, North Africa, and at lower frequencies (5% to 20%) in South and Central Asia. It is seen less frequently in North and East Asia and in the Native American populations. While admixture of Europeans in Native American populations is common, our studies overall show very low frequencies in our specific population samples except for the Maya sample (Fig. S4). Given the evidence of the variant in Northern Asia, the likely ancestral region for Native Americans, it is possible that the existence of the promoter variant at a low frequency in Native Americans is ancestral and not due to recent admixture. The same possibility applies to the presence in Australian Aborigines. The subset of 39 less admixed Australian Aborigines have a 15% frequency compared to a frequency of 40% in the full sample of 102 Aborigines. Figure 4 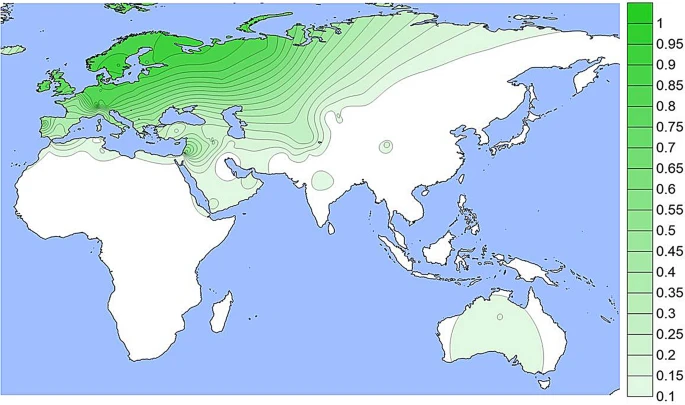 A density plot of the frequencies of the derived allele at rs12913832. The scale has been adjusted to minimize visual extrapolation to very rare occurrences. See Fig. S4, caption for Fig. 1, and text. SNPs rs1800401 and rs121918166 have not been studied in as many populations as any of the four other SNPs and we have not considered them in this study. The variant at rs121918166 has only been observed at rare frequencies in Scandinavians. Based on the populations in the 1 KG the derived allele at rs1800401 occurs most frequently, 10% to 20%, in African and South Asian populations and is absent to < 12% in East Asia and Europe. Evidence argues that the variant alleles at the four common SNPs depicted in Figs. 1, 2, 3 and 4 are functional2,8,9,11,17,24. Each of the four variants has a distinct geographic distribution but overlaps exist. In East Asia the hypomorphic rs74653330 allele overlaps somewhat with the rs1800414 variant but they appear to occur on separate haplotypes in the population. However, both the enhancer variant at rs12913832 and the amino acid substitution at rs1800407 occur frequently in Europe and surrounding areas and occur on the same chromosome at some unclear frequency. |
|
|
|
Post by Admin on Jan 19, 2021 21:10:53 GMT
Two SNP haplotype–rs1800407 and rs12913832
The interaction between the rs12913832 and rs1800407 loci is interesting. The variant allele at rs1800407 has been included in the equations used for eye color prediction11 for nearly a decade and was suggested by Sturm et al.7 as functioning to increase the penetrance of the enhancer variant. Duffy et al.29 notes that heterozygosity for the derived allele at rs1800407 decreases the probability of green eyes on the homozygous derived rs12913832 background but increases it on a heterozygous rs12913832 background. Several studies have referred to the relationship of rs1800407, especially the 419Gln allele, and the enhancer variant as an example of epistasis29,30,31,32. However, if we consider the functional unit as production of a protein we necessarily include the rate of production of mRNA and the coding content of that mRNA. The term epistasis seems inappropriate because these two DNA variants are not functionally independent loci. The haplotype is the functional unit and the locus can be considered as a four-allele locus, at least with respect to the enhancer and rs1800407 (Table 2). The phenotypes determined by three of the alleles (haplotypes) are clear; the fourth is not clear from existing studies.
Table 2 Haplotypes of the ancestral 419Arg and derived 419Gln alleles at rs1800407 and the enhancer normal (E +) and negative (E-) alleles at rs12913832. The doubly-derived (cis) haplotype has frequency estimates of 1% to 3.6% in 14 European populations (see Table S2).
From: The distinctive geographic patterns of common pigmentation variants at the OCA2 gene
2 SNP haplotype Eye color effect
419Arg-E + CA Ancestral; dark iris color
419Gln-E + TA ?
419Arg-E- CG Light iris color
419Gln-E- TG Light iris color; Doubly-derived/cis
If the doubly-derived chromosome for rs1800407 and rs12913832 results in “higher penetrance” for light eye color, the derived allele at rs1800407 must have a functional difference. While it was not studied by Sviderskaya et al.2, an obvious implication is that it is a hypomorphic allele. These cis chromosomes would have reduced production (because of the enhancer variant) of a hypomorphic OCA2 protein (because of the 419Gln allele at rs1800407). Selection operated on some trait to increase the frequency of the enhancer variant; this cis combination of the two variants with a presumably hypomorphic protein might have been more strongly affected.
On a background of homozygosity for the enhancer (rs12913832) variant, the frequency of heterozygotes of the amino acid substitution (rs1800407) is 246/(246 + 3,039) or 7.5% in Duffy’s largely British origin population sample. Those genotypes involve one chromosome that is doubly-derived (i.e., cis) for the two variants and one that has only the enhancer variant. On a heterozygous enhancer background genotype, however, the amino acid substitution heterozygotes occur at a higher frequency of 529/(529 + 1,248) or 29.8%. Those nearly 30% of individuals are composed of both cis and trans genotypes for the two functional variants. The evidence is consistent with those two genotypes having different phenotypes as would be predicted by considering the functional context: the cis genotype has one fully normal protein at normal amounts and one variant protein produced at reduced amounts; the trans genotype has a normal protein at reduced amounts and a variant protein at normal amounts.
The proportions of the two enhancer genotypes in that study29 are not necessarily in HW proportions depending on how they were ascertained, which is not specified. In fact, the ratio of the enhancer homozygotes to heterozygotes is 1.849 which is compatible with an enhancer variant frequency of about 0.79, essentially the same as in our summary (Table S1) for Northwest Europe. However, the frequency of the amino acid substitution is not so easily estimated from these data.
By maximum likelihood the phase of the ambiguous double heterozygotes will be estimated to be partly genotypes with the derived alleles in cis if there is evidence that the cis allele exists. We find (Table 3) that direct gene counting evidence of the cis haplotype is seen primarily in northern Europeans. In those populations with the gene counting evidence for this haplotype the frequency of the cis haplotype is 3%. We note the higher frequencies are in the British, Irish, and CEU samples. Several individuals in these and other populations in northern Europe and elsewhere are double heterozygotes with phase to be estimated statistically. The uncertainties of statistical phasing make it difficult with the existing sample sizes to give exact proportions of the two relevant genotypes, cis and trans. Gene counting evidence exists for both the cis and trans chromosomes; the doubly heterozygous genotype must be apportioned statistically and that is the source of uncertainty given the small numbers of the relevant genotypes setting the expectation (Table 3).
Table 3 Observed genotype counts for rs1800407 and rs12913832 among individuals with no missing data for these two SNPs. The groups shown are primarily the subset of 105 populations in Fig. 2 from world regions (Europe/SWAsia/NAfrica/SCAsia) where double heterozygotes were observed. The cells with bold underlined values indicate definite evidence of the cis (doubly-derived) haplotype,TG; see text.
From: The distinctive geographic patterns of common pigmentation variants at the OCA2 gene
rs1800407 CC CT TT CC CT TT CC CT TT
rs12913832 AA AA AA AG AG AG GG GG GG
Population N
KSR 32 3 0 5 1 0 1 0 0 42
SOU 32 0 0 14 0 0 1 0 0 47
MHD 27 1 0 9 2 0 1 0 0 40
PLA 33 5 0 21 1 0 5 0 0 65
DRU 39 5 0 38 2 0 17 0 0 101
SRD 17 3 1 12 0 0 2 0 0 35
TCP 22 5 1 20 2 0 9 0 0 59
TRK 38 4 2 24 3 0 9 0 0 80
IRN 28 4 0 8 2 0 0 0 0 42
ADY 22 4 2 18 3 0 3 1 0 53
ASH 27 5 0 45 3 0 51 0 0 131
TSI 23 9 2 50 6 0 15 2 0 107
GRK 10 6 0 23 2 0 9 0 0 50
IBS 38 10 0 40 8 1 9 1 0 107
CHV 2 1 0 13 2 0 21 3 0 42
HGR 7 0 0 37 9 0 34 2 0 89
RUA 0 0 0 6 0 0 25 2 0 33
RUV 1 0 0 13 0 0 29 1 0 44
EAM 5 2 0 26 3 0 48 3 0 87
GBR 3 1 0 18 7 0 58 3 1 91
CEU 2 2 0 29 8 1 53 4 0 99
IRI 1 5 0 21 13 0 67 7 0 114
DAN 1 1 0 8 1 0 39 1 0 51
FIN 1 0 0 8 1 0 23 1 0 34
FN1 0 0 0 16 2 0 78 3 0 99
KMZ 2 0 0 15 0 0 28 0 0 45
PTH 27 1 2 6 1 0 2 0 0 39
PJL 72 7 0 14 1 0 2 0 0 96
STU 93 3 0 5 1 0 0 0 0 102
BEB 65 4 0 15 2 0 0 0 0 86
HZR 65 11 2 12 3 0 0 0 0 93
AKZ 45 2 0 13 1 0 1 0 0 62
MGL 87 1 0 10 2 0 0 0 0 100
MAY 42 1 0 4 3 0 0 0 0 50
|
|
|
|
Post by Admin on Jan 20, 2021 20:36:18 GMT
There are 10 genotypes possible for the four haplotypes of the rs12913832 and rs1800407 variants. How all of those genotypes relate to phenotypes under selection is not known. The haplotype frequency distribution bar plots of the two SNPs common in Europe, Southwest Asia, and North Africa among 105 populations are shown in Fig. 5. (The haplotype frequencies are in supplemental Table S2). In our data we have seen direct evidence for 8 of those genotypes (Table 4). The variants at rs12913832 and rs1800407 occur in cis at the highest frequencies in the northern European samples and the gene-counting evidence for the cis chromosome occurs almost exclusively in these northern European populations (Table 3). These haplotypes are relevant to how genotypes might influence pigmentation and selection in those northern European populations. While random genetic drift can always be a possible explanation for the pattern, it seems a highly unlikely explanation for the evidence of this doubly-derived chromosome to exist only in the populations for which evidence of selection on the enhancer chromosomes is strongest. Figure 5 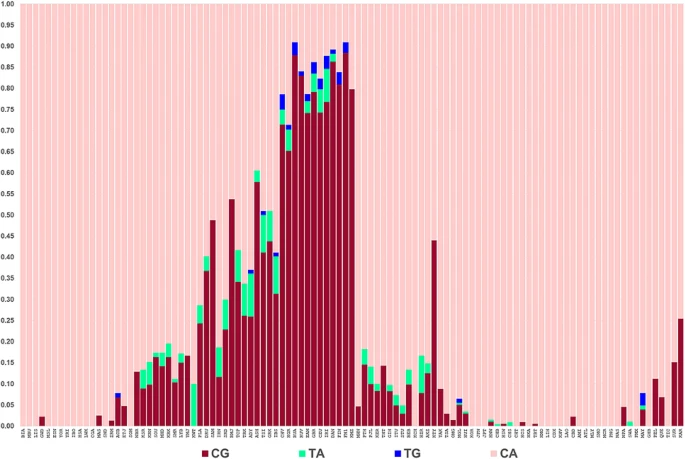 The haplotypes of rs12913832 and rs1800407 showing the high relative frequency of the doubly-derived haplotype especially in Northern Europe.
Table 4 Distribution of individuals (by direct gene counting and by inference) in 105 populations for the 10 possible genotypes of the 2-SNP haplotype based on rs1800407, rs12913832.
From: The distinctive geographic patterns of common pigmentation variants at the OCA2 gene
Ten possible genotypes Direct counting Inferred by PHASE from double heterozygotes Inferred missing genotype Total individuals Observed in populations
CA CA 4,643 0 179 4,822
CA CG 885 0 40 925
CG CG 676 0 18 694
CA TA 131 0 6 137
CG TA 0 87 0 87
CG TG 35 0 0 35
TA TA 13 0 1 14
CA TG 0 8 0 8
TA TG 2 0 0 2 CEU, IBS
TG TG 1 0 0 1 GBR
Total 6,386 95 244 6,725
We can expect, given the functional variation at each site, that all but the homozygote for the doubly ancestral genotype will have some positive effect toward lighter pigmentation. However, evidence for the effect on eye color of just the variant (419Gln) as heterozygous with a fully ancestral chromosome is largely absent; its expected frequency is quite small. Even assuming the haplotypes affect phenotype additively, to estimate the three different fitness parameters associated with the three derived chromosomes seems beyond the power of the existing data. We leave such estimation to others. Many studies have reported on use of the genotypes at these sites at OCA2 and SNPs at other genes to infer the iris, hair, and skin color of an individual from that individual’s DNA. Those efforts are most recently integrated into the HIrisplex-S web site (https://hirisplex.erasmusmc.nl/). Such phenotype inference from a DNA sample can be very useful as an investigative lead in criminal forensics. Our data summaries demonstrate that two of the SNPs, rs12913832 and rs1800414, have common variants with strikingly different geographic patterns that makes them relevant to inference of biogeographic ancestry in some parts of the world. Indeed, rs12913832, the enhancer SNP, was incorporated in the Kidd Lab panel of 55 ancestry informative SNPs26 and rs1800414 is part of the 74 SNPs in a panel by Li et al.33. 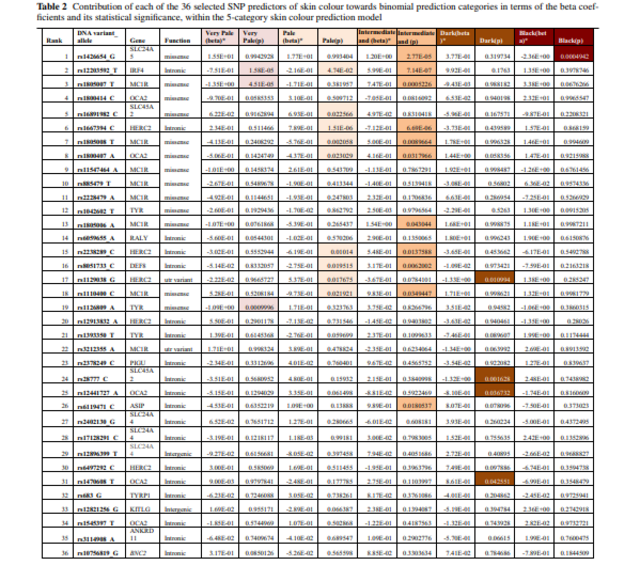 The population distribution of the chromosome with the derived enhancer variant (rs12913832) and the derived amino acid variant (419Gln for rs1800407) in cis is seen almost exclusively in northern Europe. Elsewhere, the rs1800407 variant (419Gln) occurs on a chromosome with the ancestral allele at the enhancer. The common occurrence of the doubly-derived (cis) chromosome, primarily in the populations with the strongest evidence of selection for the enhancer variant, strongly suggests selection on this chromosome in northern Europe. The north Eurasia distribution of the hypomorphic allele–481Thr at rs74653330–suggests parallel evolution for this variant as well. Our understanding of the role of the known functional and enhancer variants in human pigmentation phenotypes has grown markedly in recent decades but, thus far, the relationships have only been studied simultaneously and in relatively large samples in a subset of populations of European and East Asian ancestry. The very strong geographical frequency patterns shown by the existing patchwork of genetic data in the OCA2-HERC2 gene region are more extensive and suggest that more empirical studies are needed from more world regions so that we can refine and improve our knowledge. The studies supporting strong selection effects done thus far also support the view that more studies are important. Other genetic loci are known to influence pigmentation phenotypes. Their relative roles and the magnitude of their effects during development as well as the evolutionary impact of non-genetic factors will be more clearly understood when we have more worldwide data on the OCA2-HERC2 gene region. Informed consent All subjects gave permission for collection of samples and use in population studies such as this. All samples are anonymous. |
|
|
|
Post by Admin on Jan 25, 2021 20:59:32 GMT
Adaptation of human skin color in various populations Lian Deng & Shuhua Xu Hereditas volume 155, Article number: 1 (2018) Abstract Background Skin color is a well-recognized adaptive trait and has been studied extensively in humans. Understanding the genetic basis of adaptation of skin color in various populations has many implications in human evolution and medicine. Discussion Impressive progress has been made recently to identify genes associated with skin color variation in a wide range of geographical and temporal populations. In this review, we discuss what is currently known about the genetics of skin color variation. We enumerated several cases of skin color adaptation in global modern humans and archaic hominins, and illustrated why, when, and how skin color adaptation occurred in different populations. Finally, we provided a summary of the candidate loci associated with pigmentation, which could be a valuable reference for further evolutionary and medical studies. Conclusion Previous studies generally indicated a complex genetic mechanism underlying the skin color variation, expanding our understanding of the role of population demographic history and natural selection in shaping genetic and phenotypic diversity in humans. Future work is needed to dissect the genetic architecture of skin color adaptation in numerous ethnic minority groups around the world, which remains relatively obscure compared with that of major continental groups, and to unravel the exact genetic basis of skin color adaptation. Background Since modern humans ventured out of Africa ~100,000 years ago, they spread across continents into a variety of habitats, from tropical zones to the arctic, and from lowlands to highlands. During migration, selective pressures in local environments (e.g., the cold climate, hypoxia, and endemic pathogens), together with random drift, have resulted in population-specific genetic variants, which further influenced variable phenotypes, such as lactose tolerance, height, immune system, and metabolic efficiency. Skin color variation is one of the most striking examples of human phenotypic diversity. It is dominated by melanin, a pigmentation located in the base of the epidermis and produced by melanocytes. Melanin has two forms, pheomelanin (yellow-reddish) and eumelanin (black-brown). The former is mainly accumulated in the light-complexioned people, while the latter is mostly produced in the dark-complexioned people [1,2,3,4,5]. In addition, the number and size of melanin particles differ among individuals, and is even more important than the proportions of the two forms of melanin in the determination of human skin color [5]. Other skin-related factors, e.g., keratin, also contribute to skin color variation [6, 7]. In global populations, skin color is highly correlated with latitude, and fundamentally, the distribution of ultraviolet (UV) radiation (Fig. 1). Populations closer to the equator tend to have dark skin for protection against UV, since overexposure to UV may decrease folic acid levels [8, 9] and cause skin cancer [10,11,12,13]. The lighter skin in populations at higher latitudes is underlying selection to maintain vitamin D photosynthesis, which is a UV-dependent process [14, 15]. Fig. 1 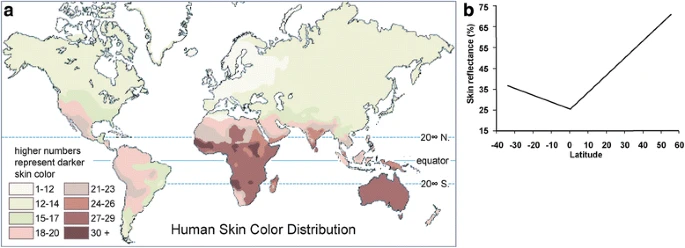 Correlation between skin color and latitude (from Barsh (2003) [5]). (a) A map of human skin color distribution. (b) A plot of skin reflectance against latitude Although UV has been assumed to be a driving force for the evolution of human skin colors, understanding the exact genetic mechanism of selection would be crucial to reconstruct human evolutionary history and elucidate the microevolution of adaptive traits. Describing a full picture of regional skin color adaptation in humans would be challenging because it includes not only the genes identified to be under selection, but also the extent to which these genes could explain phenotypic variation, the interactions and joint effects of genes, and the way they react to the external environments. In this article, we reviewed several cases of skin color adaptation in various populations of modern humans and archaic hominins. These cases show the similarities and differences of mechanisms of skin color adaptation across populations, and provide some insights into human evolutionary history. |
|









