|
|
Post by Admin on Feb 5, 2019 17:54:55 GMT
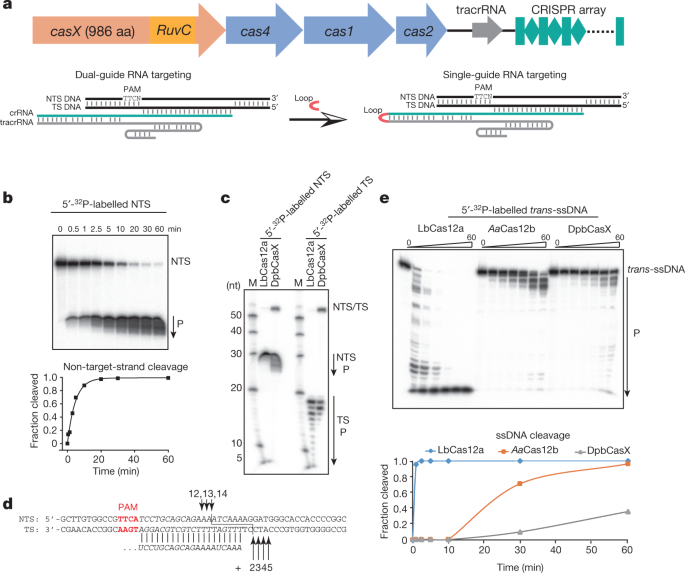 In a mere seven years, Cas9 has shown itself to be a formidable gene editor, employed in humans, plants, animals and bacteria to quickly and accurately cut and splice DNA, transforming biology and opening new avenues for treating disease. Discovered two years ago by UC Berkeley scientists Jill Banfield and Jennifer Doudna in some of the world's smallest bacteria, the protein was similar to Cas9, but quite a bit smaller: a big advantage if you're trying to deliver a gene editor into a cell.  According to a study published today in Nature, CasX is, in fact, a potent and efficient gene editor in both bacteria and human cells. Its design is similar to Cas9 and its well-studied cousin, Cas12, but it is different enough that it appears to have evolved in bacteria independently of the other Cas proteins. It can cut double-stranded DNA like Cas9, can bind to DNA to regulate genes, and it can be targeted to specific DNA sequences like other Cas proteins. Plus, because it comes from bacteria that are not found in humans—Banfield dredged them from a database of microbes found in groundwater and sediment—the human immune system should accept it more easily than Cas9. Some doctors fear that Cas9 may create an immune reaction in patients treated with CRISPR therapies. 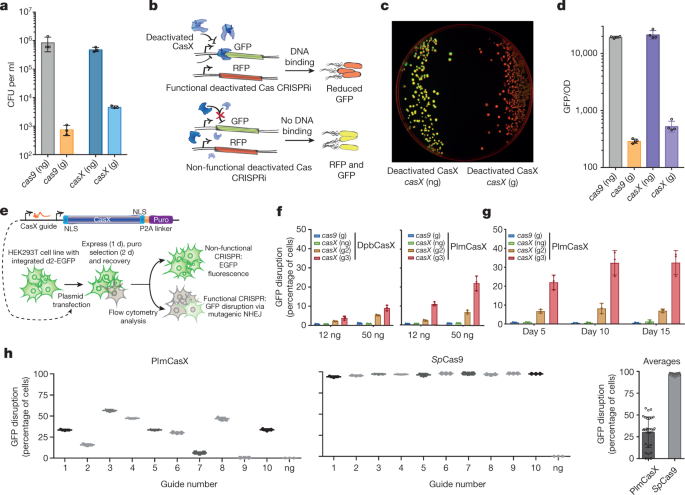 "The immunogenicity, delivery and specificity of a genome-editing tool are all vitally important," said co-lead author Benjamin Oakes, a former UC Berkeley graduate student and current Entrepreneurial Fellow in the Innovative Genomics Institute. "We're excited about CasX on all of these fronts." The RNA-guided CRISPR-associated (Cas) proteins Cas9 and Cas12a provide adaptive immunity against invading nucleic acids, and function as powerful tools for genome editing in a wide range of organisms. Here we reveal the underlying mechanisms of a third, fundamentally distinct RNA-guided genome-editing platform named CRISPR–CasX, which uses unique structures for programmable double-stranded DNA binding and cleavage. Biochemical and in vivo data demonstrate that CasX is active for Escherichia coli and human genome modification. Eight cryo-electron microscopy structures of CasX in different states of assembly with its guide RNA and double-stranded DNA substrates reveal an extensive RNA scaffold and a domain required for DNA unwinding. These data demonstrate how CasX activity arose through convergent evolution to establish an enzyme family that is functionally separate from both Cas9 and Cas12a. 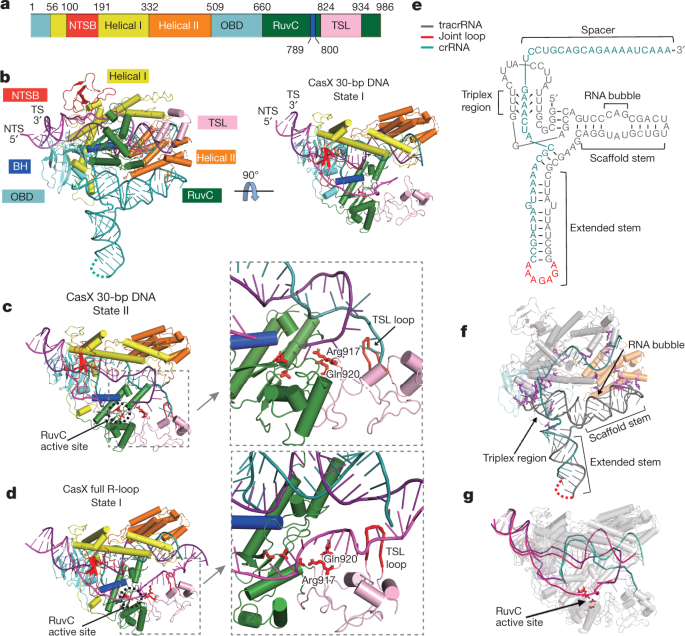 Overall structure of the CasX ternary complex. |
|
|
|
Post by Admin on Feb 7, 2019 17:49:41 GMT
Officials at Stanford University have opened an investigation into what several high-profile faculty members knew about a Chinese effort to create gene-edited babies led by a onetime researcher at the California school, He Jiankui. The investigation, according to people familiar with it, aims to understand what liabilities or risks Stanford may have in connection with the controversial medical experiment, which led last year to the birth of two girls whose genomes had been altered with a molecular tool called CRISPR to render them immune to HIV.  In an e-mail, Stanford confirmed the inquiry. “We have a review under way of the circumstances around Dr. He’s interactions with researchers at the university,” said spokesperson Ernest Miranda. In November, MIT Technology Review reported that He, by then a professor at the Southern University of Science and Technology, in Shenzhen, had begun a daring and ethically charged effort to create the first CRISPR babies. He, who claimed twins girls named Lulu and Lala had been born, was immediately placed under investigation by Chinese authorities and could face serious legal consequences. |
|
|
|
Post by Admin on Apr 14, 2019 18:40:28 GMT
Genetic engineering, being a direct manipulation of a living organism's genes, have advanced as it has faced many ethical issues.  Just recently, a group of Chinese scientists has implanted human brain genes into 11 rhesus monkeys. The study, as the team explains, is intended to provide a different perspective on the evolution of human intelligence.  Human versions of the MCPH1 gene was used in the study. The said gene is supposedly in control when it comes to human brain development.  In their study, the rhesus monkeys were found to have taken a longer period in brain development, much like humans. The subject monkeys were also said to have performed in tests better than the unmodified subjects. The tests, focusing on short-term memory and reaction time, have resulted in five monkeys doing well and getting a passing mark. The memory test entails remembering colors and shapes on a screen. Later, the subject monkeys were given MRI scans. This test done by China has yet again fueled debates on ethics as with their previous biomedical experiment. The ethical concerns have risen because of comparisons with "Planet of the Apes," a dystopian sci-fi film. The research was conducted by a team at the Kunming Institute of Zoology and the Chinese Academy of Sciences. They are working in collaboration with a team of US researchers from the University of North Carolina. Abstract Brain size and cognitive skills are the most dramatically changed traits in humans during evolution, and yet the genetic mechanisms underlying these human-specific changes remain elusive. Here, we successfully generated 11 transgenic rhesus monkeys (8 first-generation and 3 second-generation) carrying human copies of MCPH1, an important gene for brain development and brain evolution. Brain image and tissue section analyses indicated an altered pattern of neural cell differentiation, resulting in a delayed neuronal maturation and neural fiber myelination of the transgenic monkeys, similar to the known evolutionary change of developmental delay (neoteny) in humans. Further brain transcriptome and tissue section analyses of major developmental stages showed a marked human-like expression delay of neuron-differentiation and synaptic signaling genes, providing a molecular explanation to the observed brain developmental delay of the transgenic monkeys. More importantly, the transgenic monkeys exhibited better short-term memory and shorter reaction time compared to the wild type controls in the delayed matching to sample task. The presented data represents the first attempt to experimentally interrogate the genetic basis of human brain origin using a transgenic monkey model, and it values the use of nonhuman primates in understanding human unique traits. National Science Review, nwz043, doi.org/10.1093/nsr/nwz043Published: 27 March 2019 |
|








