Post by Admin on Apr 14, 2023 17:13:23 GMT
Treating seizures and epilepsy with anticoagulants?
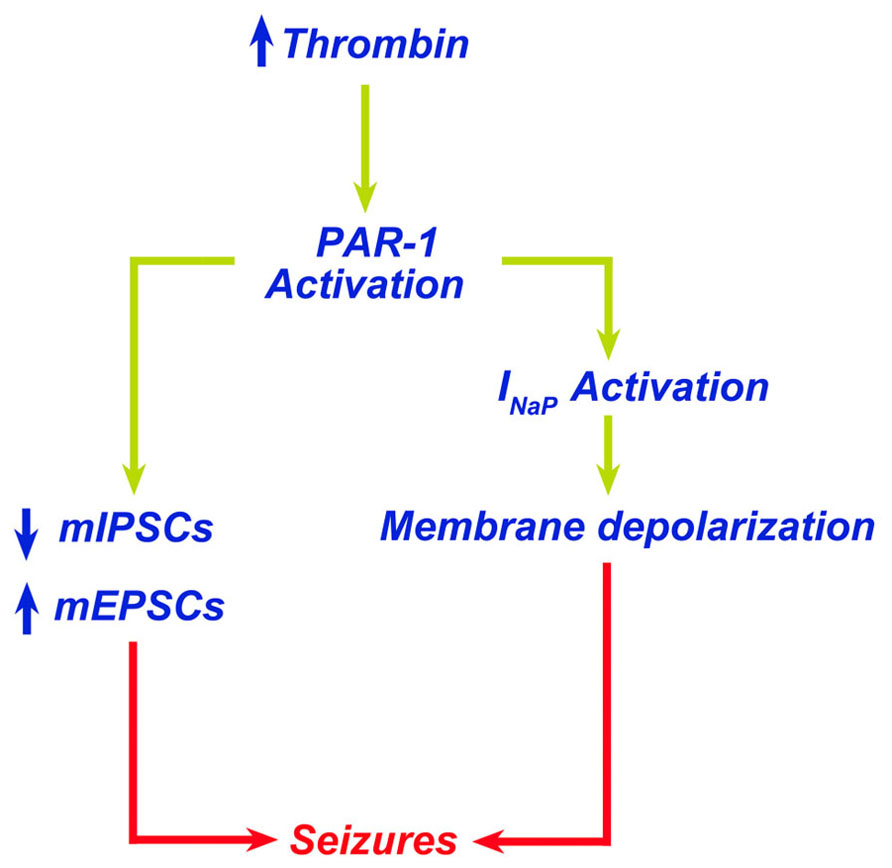
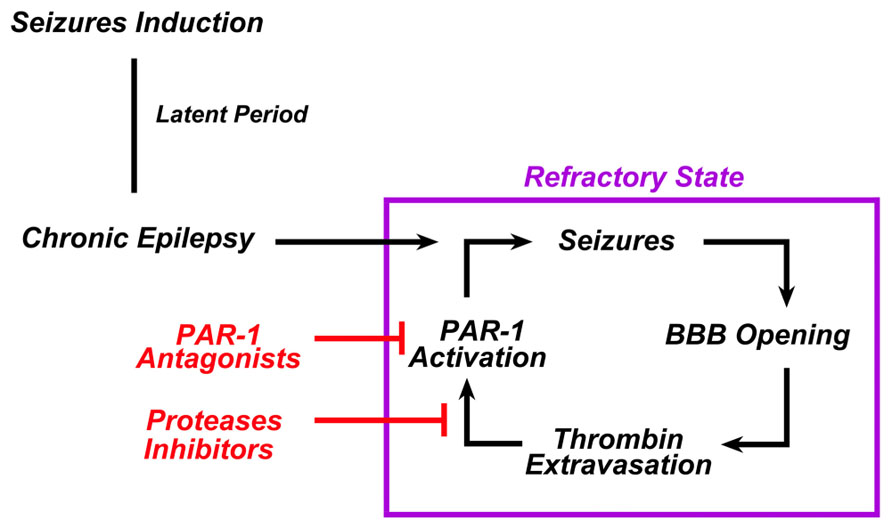
Thrombin is a serine protease playing an essential role in the blood coagulation cascade. Recent work, however, has identified a novel role for thrombin-mediated signaling pathways in the central nervous system. Binding of thrombin to protease-activated receptors (PARs) in the brain appears to have multiple actions affecting both health and disease. Specifically, thrombin has been shown to lead to the onset of seizures via PAR-1 activation. In this perspective article, we review the putative mechanisms by which thrombin causes seizures and epilepsy. We propose a potential role of PAR-1 antagonists and novel thrombin inhibitors as new, possible antiepileptic drugs.
Thrombin Signaling in the Brain
Thrombin is a serine protease, which plays an essential role in the blood coagulation cascade (Siller-Matula et al., 2011). Upon its formation following the enzymatic cleavage of prothrombin by activated Factor X, thrombin regulates a cascade of proteolytic events ultimately leading to the formation of blood clots (Lippi et al., 2012). Lately, however, novel signaling cascades mediated by thrombin have been discovered (Siller-Matula et al., 2011). Specifically, through the activation of the protease-activated receptors (PARs), thrombin seems to directly affect the activity of multiple cell types and regulate a variety of biological functions, such as inflammation, leukocyte migration, cellular proliferation, vascular permeability and tone, edema formation, and other processes related to tissue repair (Coughlin, 2000, 2001; Sambrano et al., 2001; Chen and Dorling, 2009; Schuepbach et al., 2009; Spiel et al., 2011).
Protease-activated receptors belong to a unique family of G protein-coupled receptors (Luo et al., 2007). Their activation is initiated by an irreversible site-specific proteolytic cleavage in the N-terminal extracellular region. The uncovered N-terminal region then acts as a tethered ligand which activates the receptor (Gingrich and Traynelis, 2000). PARs are expressed in the brain and while PAR-2 represents a class of trypsin/tryptase-activated receptors, PAR-1, PAR-3, and PAR-4 are most effectively activated by thrombin (Gingrich and Traynelis, 2000). In the brain, PAR-1 has been detected in both neurons and astrocytes, with the latter demonstrating stronger immunoreactivity in human brain tissue (Junge et al., 2004). High levels of PAR-1 are detected in the hippocampus, cortex, and striatum of humans (Junge et al., 2004). While the molecular pathways activated by PAR-1 in neurons are yet under investigation, in the brain PAR-1 activation has been shown to modulate synaptic transmission and plasticity through the enhancement of N-methyl-D-aspartate (NMDA) receptor (NMDAR) currents (Gingrich et al., 2000; Lee et al., 2007; Maggio et al., 2008). In addition, PAR-1 knockout animals present profound deficits in hippocampus-dependent learning and memory processes (Almonte et al., 2007, 2013). Altogether, it seems that PAR-1 plays a critical role in memory formation and synaptic plasticity.
Interestingly, a variety of pathological conditions have been associated with changes in the expression of PAR-1 in the brain. In Parkinson's disease, a significant increase in the number of astrocytes expressing PAR-1 has been reported in the substantia nigra pars compacta (Ishida et al., 2006). In addition, upregulation of PAR-1 in astrocytes has been observed in HIV encephalitis, (Boven et al., 2003) indicating that this receptor might be implicated in the pathogenesis of neuroinflammation. This idea is supported by the evidence of elevated levels of thrombin in an experimental model of multiple sclerosis (Beilin et al., 2005) as well as in other inflammatory brain diseases (Chapman, 2006). Stimulation of PAR-1 by thrombin causes proliferation of glia and potentially produces reactive gliosis, infiltration of inflammatory cells, and angiogenesis (Striggow et al., 2001). Finally, expression of PAR-1 is increased in experimental models of Alzheimer's disease (Pompili et al., 2004) and brain ischemia (Striggow et al., 2001).
www.frontiersin.org/articles/10.3389/fncel.2013.00019/full
The critical interaction between valproate sodium and warfarin: case report and review
Abstract
Background
Valproic acid (VPA) and warfarin are commonly prescribed for patients with epilepsy and concomitant atrial fibrillation (AF). When VPA and warfarin are prescribed together, clinically important interactions may occur. VPA may replace warfarin from the protein binding sites and result in an abnormally increased anticoagulation effect. This is commonly underrecognized.
Case presentation
In our case, we report a 78-year-old woman with a glioma who presented with status epilepticus. The patient was on warfarin to prevent cardiogenic embolism secondary to AF. Intravenous loading dose of VPA was administered, but international normalized ratio (INR) increased significantly to 8.26. Intravenous vitamin K1 was then given and the patient developed no overt bleeding during the hospitalization.
Conclusion
By reviewing the literature and discussing the critical interaction between valproate sodium and warfarin, we conclude that intravenous VPA and the co-administrated warfarin may develop critical but underrecognized complications due to effects on the function of hepatic enzymes and displacement of protein binding sites.
Keywords: Valproic acid, Warfarin, Interaction, International normalized ratio, Epilepsy
www.ncbi.nlm.nih.gov/pmc/articles/PMC6167846/
In a recent CMAJ commentary on drug interactions with warfarin,1 David Juurlink overlooked a very important category of drugs that interact with warfarin: antiepileptic drugs. Phenytoin, carbamazepine and phenobarbital are potent inducers of the cytochrome P450 system, and their interactions with warfarin have been known for decades.2–5 These drugs can substantially increase the rate at which warfarin is metabolized and thus reduce the effect of a previously adjusted dose. Likewise, sudden withdrawal of any of these drugs may decrease the rate at which warfarin is metabolized and put a patient taking a combination of these drugs at an increased risk of bleeding. Antiepileptic drugs are not only prescribed for epilepsy, which is estimated to affect 200 000 Canadians; they are also used to treat psychiatric disorders in a large population of patients. I suggest that Juurlink should add antiepileptic drugs as a ninth category to Table 1.1
REFERENCES
1. Juurlink DN. Drug interactions with warfarin: what clinicians need to know. CMAJ 2007;177:369-71. [PMC free article] [PubMed]
2. Herman D, Locatelli I, Grabnar I, et al. The influence of co-treatment with carbamazepine, amiodarone and statins on warfarin metabolism and maintenance dose. Eur J Clin Pharmacol 2006;62:291-6. [PubMed]
3. Solomom GE, Hilgartner MW, Kutt H. Coagulation defects caused by diphenylhydantoin. Neurology 1972;22:1165-71. [PubMed]
4. Nappi JM. Warfarin and phenytoin interactions. Ann Intern Med 1979;90:852. [PubMed]
5. Udall JA. Clinical implications of warfarin interactions with 5 sedatives. Am J Cardiol 1975;35:67-71.
[PubMed]
Novel vitamin K analogue eliminates seizure activity in epileptic mouse models
Researchers report that their novel vitamin K-based drug prevented seizures in murine models of medication-resistant epilepsy. The team highlighted that the drug is orally bioavailable, has excellent brain penetration and rapidly distributes through the central nervous system.
The research at the Medical University of South Carolina (MUSC), US, was led by Dr Sherine Chan and Dr James Chou, both associate professors in the Department of Drug Discovery and Biomedical Sciences in MUSC’s College of Pharmacy, and the co-founders of Neuroene Therapeutics, a startup company aiming to develop therapies for medication-resistant conditions caused by mitochondrial dysfunction, such as epilepsy.
According to the group, approximately a third of US epilepsy patients live with a medication-resistant type, where uncontrolled and sudden seizures put them at higher risk of injury.

Despite the availability of more than 25 antiseizure drugs on the market, approximately 30% of patients with epilepsy still suffer from seizures. Thus, the epilepsy therapy market has a great need for a breakthrough drug that will aid pharmacoresistant patients. In our previous study, we discovered a vitamin K analogue, 2h, which displayed modest antiseizure activity in zebrafish and mouse seizure models. However, there are limitations to this compound due to its pharmacokinetic profile. In this study, we develop a new series of vitamin K analogues by modifying the structure of 2h. Among these, compound 3d shows full protection in a rodent pharmacoresistant seizure model with limited rotarod motor toxicity and favorable pharmacokinetic properties. Furthermore, the brain/plasma concentration ratio of 3d indicates its excellent permeability into the brain. The resulting data shows that 3d can be further developed as a potential antiseizure drug in the clinic.
In their paper, the team developed a modified form of naturally occurring vitamin K as a novel seizure prevention drug. According to the Chan, the therapeutic eliminated seizure activity in all the murine models of epilepsy it was used on and was well tolerated in both mice and rats.
The successful drug was developed alongside 22 other vitamin K analogues, all of which were tested for their effectiveness in controlling seizures in different epilepsy types in mice. The authors believe it is the unique structure they have designed for this one molecule that makes it effective in controlling medication-resistant seizures.
Epilepsy can be caused by mitochondrial dysfunction in brain cells. Mitochondria produce most of the energy for the cell and so dysfunction affects the supply of energy for cellular processes; because brain cells require a significant amount of energy to function, changes to mitochondrial output disrupts their function, Chan explained. She added: “This dysfunction is an underlying cause of many neurological diseases, including epilepsy, Parkinson’s disease and rare mitochondrial disorders.”
In the study, treatment with the vitamin K analogue increased the ability of the brain cells to produce oxygen. It is believed by the researchers that this could be a key feature of the drug’s mechanism of action in controlling seizures.
The study tested the drug in mouse brain cells, a zebra fish seizure model and multiple mouse seizure models representing different types of epilepsy. According to Chou, the team were thrilled that after the drug showed promise in the early cell and zebrafish models, it eliminated seizure activity in all mice tested.
pubs.acs.org/doi/10.1021/acs.jmedchem.0c00168
Thrombin is a serine protease playing an essential role in the blood coagulation cascade. Recent work, however, has identified a novel role for thrombin-mediated signaling pathways in the central nervous system. Binding of thrombin to protease-activated receptors (PARs) in the brain appears to have multiple actions affecting both health and disease. Specifically, thrombin has been shown to lead to the onset of seizures via PAR-1 activation. In this perspective article, we review the putative mechanisms by which thrombin causes seizures and epilepsy. We propose a potential role of PAR-1 antagonists and novel thrombin inhibitors as new, possible antiepileptic drugs.
Thrombin Signaling in the Brain
Thrombin is a serine protease, which plays an essential role in the blood coagulation cascade (Siller-Matula et al., 2011). Upon its formation following the enzymatic cleavage of prothrombin by activated Factor X, thrombin regulates a cascade of proteolytic events ultimately leading to the formation of blood clots (Lippi et al., 2012). Lately, however, novel signaling cascades mediated by thrombin have been discovered (Siller-Matula et al., 2011). Specifically, through the activation of the protease-activated receptors (PARs), thrombin seems to directly affect the activity of multiple cell types and regulate a variety of biological functions, such as inflammation, leukocyte migration, cellular proliferation, vascular permeability and tone, edema formation, and other processes related to tissue repair (Coughlin, 2000, 2001; Sambrano et al., 2001; Chen and Dorling, 2009; Schuepbach et al., 2009; Spiel et al., 2011).
Protease-activated receptors belong to a unique family of G protein-coupled receptors (Luo et al., 2007). Their activation is initiated by an irreversible site-specific proteolytic cleavage in the N-terminal extracellular region. The uncovered N-terminal region then acts as a tethered ligand which activates the receptor (Gingrich and Traynelis, 2000). PARs are expressed in the brain and while PAR-2 represents a class of trypsin/tryptase-activated receptors, PAR-1, PAR-3, and PAR-4 are most effectively activated by thrombin (Gingrich and Traynelis, 2000). In the brain, PAR-1 has been detected in both neurons and astrocytes, with the latter demonstrating stronger immunoreactivity in human brain tissue (Junge et al., 2004). High levels of PAR-1 are detected in the hippocampus, cortex, and striatum of humans (Junge et al., 2004). While the molecular pathways activated by PAR-1 in neurons are yet under investigation, in the brain PAR-1 activation has been shown to modulate synaptic transmission and plasticity through the enhancement of N-methyl-D-aspartate (NMDA) receptor (NMDAR) currents (Gingrich et al., 2000; Lee et al., 2007; Maggio et al., 2008). In addition, PAR-1 knockout animals present profound deficits in hippocampus-dependent learning and memory processes (Almonte et al., 2007, 2013). Altogether, it seems that PAR-1 plays a critical role in memory formation and synaptic plasticity.
Interestingly, a variety of pathological conditions have been associated with changes in the expression of PAR-1 in the brain. In Parkinson's disease, a significant increase in the number of astrocytes expressing PAR-1 has been reported in the substantia nigra pars compacta (Ishida et al., 2006). In addition, upregulation of PAR-1 in astrocytes has been observed in HIV encephalitis, (Boven et al., 2003) indicating that this receptor might be implicated in the pathogenesis of neuroinflammation. This idea is supported by the evidence of elevated levels of thrombin in an experimental model of multiple sclerosis (Beilin et al., 2005) as well as in other inflammatory brain diseases (Chapman, 2006). Stimulation of PAR-1 by thrombin causes proliferation of glia and potentially produces reactive gliosis, infiltration of inflammatory cells, and angiogenesis (Striggow et al., 2001). Finally, expression of PAR-1 is increased in experimental models of Alzheimer's disease (Pompili et al., 2004) and brain ischemia (Striggow et al., 2001).
www.frontiersin.org/articles/10.3389/fncel.2013.00019/full
The critical interaction between valproate sodium and warfarin: case report and review
Abstract
Background
Valproic acid (VPA) and warfarin are commonly prescribed for patients with epilepsy and concomitant atrial fibrillation (AF). When VPA and warfarin are prescribed together, clinically important interactions may occur. VPA may replace warfarin from the protein binding sites and result in an abnormally increased anticoagulation effect. This is commonly underrecognized.
Case presentation
In our case, we report a 78-year-old woman with a glioma who presented with status epilepticus. The patient was on warfarin to prevent cardiogenic embolism secondary to AF. Intravenous loading dose of VPA was administered, but international normalized ratio (INR) increased significantly to 8.26. Intravenous vitamin K1 was then given and the patient developed no overt bleeding during the hospitalization.
Conclusion
By reviewing the literature and discussing the critical interaction between valproate sodium and warfarin, we conclude that intravenous VPA and the co-administrated warfarin may develop critical but underrecognized complications due to effects on the function of hepatic enzymes and displacement of protein binding sites.
Keywords: Valproic acid, Warfarin, Interaction, International normalized ratio, Epilepsy
www.ncbi.nlm.nih.gov/pmc/articles/PMC6167846/
In a recent CMAJ commentary on drug interactions with warfarin,1 David Juurlink overlooked a very important category of drugs that interact with warfarin: antiepileptic drugs. Phenytoin, carbamazepine and phenobarbital are potent inducers of the cytochrome P450 system, and their interactions with warfarin have been known for decades.2–5 These drugs can substantially increase the rate at which warfarin is metabolized and thus reduce the effect of a previously adjusted dose. Likewise, sudden withdrawal of any of these drugs may decrease the rate at which warfarin is metabolized and put a patient taking a combination of these drugs at an increased risk of bleeding. Antiepileptic drugs are not only prescribed for epilepsy, which is estimated to affect 200 000 Canadians; they are also used to treat psychiatric disorders in a large population of patients. I suggest that Juurlink should add antiepileptic drugs as a ninth category to Table 1.1
REFERENCES
1. Juurlink DN. Drug interactions with warfarin: what clinicians need to know. CMAJ 2007;177:369-71. [PMC free article] [PubMed]
2. Herman D, Locatelli I, Grabnar I, et al. The influence of co-treatment with carbamazepine, amiodarone and statins on warfarin metabolism and maintenance dose. Eur J Clin Pharmacol 2006;62:291-6. [PubMed]
3. Solomom GE, Hilgartner MW, Kutt H. Coagulation defects caused by diphenylhydantoin. Neurology 1972;22:1165-71. [PubMed]
4. Nappi JM. Warfarin and phenytoin interactions. Ann Intern Med 1979;90:852. [PubMed]
5. Udall JA. Clinical implications of warfarin interactions with 5 sedatives. Am J Cardiol 1975;35:67-71.
[PubMed]
Novel vitamin K analogue eliminates seizure activity in epileptic mouse models
Researchers report that their novel vitamin K-based drug prevented seizures in murine models of medication-resistant epilepsy. The team highlighted that the drug is orally bioavailable, has excellent brain penetration and rapidly distributes through the central nervous system.
The research at the Medical University of South Carolina (MUSC), US, was led by Dr Sherine Chan and Dr James Chou, both associate professors in the Department of Drug Discovery and Biomedical Sciences in MUSC’s College of Pharmacy, and the co-founders of Neuroene Therapeutics, a startup company aiming to develop therapies for medication-resistant conditions caused by mitochondrial dysfunction, such as epilepsy.
According to the group, approximately a third of US epilepsy patients live with a medication-resistant type, where uncontrolled and sudden seizures put them at higher risk of injury.

Despite the availability of more than 25 antiseizure drugs on the market, approximately 30% of patients with epilepsy still suffer from seizures. Thus, the epilepsy therapy market has a great need for a breakthrough drug that will aid pharmacoresistant patients. In our previous study, we discovered a vitamin K analogue, 2h, which displayed modest antiseizure activity in zebrafish and mouse seizure models. However, there are limitations to this compound due to its pharmacokinetic profile. In this study, we develop a new series of vitamin K analogues by modifying the structure of 2h. Among these, compound 3d shows full protection in a rodent pharmacoresistant seizure model with limited rotarod motor toxicity and favorable pharmacokinetic properties. Furthermore, the brain/plasma concentration ratio of 3d indicates its excellent permeability into the brain. The resulting data shows that 3d can be further developed as a potential antiseizure drug in the clinic.
In their paper, the team developed a modified form of naturally occurring vitamin K as a novel seizure prevention drug. According to the Chan, the therapeutic eliminated seizure activity in all the murine models of epilepsy it was used on and was well tolerated in both mice and rats.
The successful drug was developed alongside 22 other vitamin K analogues, all of which were tested for their effectiveness in controlling seizures in different epilepsy types in mice. The authors believe it is the unique structure they have designed for this one molecule that makes it effective in controlling medication-resistant seizures.
Epilepsy can be caused by mitochondrial dysfunction in brain cells. Mitochondria produce most of the energy for the cell and so dysfunction affects the supply of energy for cellular processes; because brain cells require a significant amount of energy to function, changes to mitochondrial output disrupts their function, Chan explained. She added: “This dysfunction is an underlying cause of many neurological diseases, including epilepsy, Parkinson’s disease and rare mitochondrial disorders.”
In the study, treatment with the vitamin K analogue increased the ability of the brain cells to produce oxygen. It is believed by the researchers that this could be a key feature of the drug’s mechanism of action in controlling seizures.
The study tested the drug in mouse brain cells, a zebra fish seizure model and multiple mouse seizure models representing different types of epilepsy. According to Chou, the team were thrilled that after the drug showed promise in the early cell and zebrafish models, it eliminated seizure activity in all mice tested.
pubs.acs.org/doi/10.1021/acs.jmedchem.0c00168