|
|
Post by Admin on Jul 6, 2023 21:46:46 GMT
In 2014, more human fossil remains were discovered at Afontova Gora II during salvage excavation before the construction of a new bridge over the Yenesei River. The remains belonged to two different females: the atlas of an adult female and the mandible and five lower teeth of a teenage girl (Afontova Gora 3) estimated to be around 14–15 years old. In 2017, direct AMS dating revealed that Afontova Gora 3 is dated to around 16,090 cal BC). DNA was extracted from one of the teeth of Afontova Gora 3 and analyzed. Compared to Afontova Gora 2, researchers were able to obtain higher coverage genomes from Afontova Gora 3. DNA analysis confirmed that the individual was female. mtDNA analysis revealed that Afontova Gora 3 belonged to the mitochondrial Haplogroup R1b. Around 2.9-3.7% of the genome was Neanderthal in origin.  In a 2016 study, researchers determined that Afontova Gora 2, Afontova Gora 3, and Mal'ta 1 (Mal'ta boy) shared common descent and were clustered together in a Mal'ta cluster. Genetically, Afontova Gora 3 is not closer to Afontova Gora 2 when compared to Mal'ta 1. When compared to Mal'ta 1, the Afontova Gora 3 lineage apparently contributed more to modern humans and is genetically closer to Native Americans. The earliest known example of the classic European blond hair mutation is in an Ancient North Eurasian from the Lake Baikal region of eastern Siberia from seventeen thousand years ago. The hundreds of millions of copies of this mutation in central and western Europe today likely derive from a massive migration of people bearing Ancient North Eurasian ancestry. Phenotypic analysis shows that Afontova Gora 3 carries the derived rs12821256 allele associated with, and likely causal for, blond hair color, making Afontova Gora 3 the earliest individual known to carry this derived allele. The allele was found in three later members of the largely ANE-derived Eastern Hunter-Gatherers populations from Samara, Motala and Ukraine c. 10,000 BP, suggesting that it originated in the Ancient North Eurasian population before spreading to western Eurasia. The hundreds of millions of copies of this mutated allele (a single-nucleotide polymorphism) are at the root of the classic European blond hair mutation, as massive population migrations from the Eurasian steppe, by a people who had substantial Ancient North Eurasian ancestry, entered continental Europe.  Supplementary Information page 52: "The derived allele of the KITLG SNP rs12821256 that is associated with – and likely causal for blond hair in Europeans is present in one hunter-gatherer from each of Samara, Motala and Ukraine (I0124, I0014 and I1763), as well as several later individuals with Steppe ancestry. Since the allele is found in populations with EHG but not WHG ancestry, it suggests that its origin is in the Ancient North Eurasian (ANE) population. Consistent with this, we observe that the earliest known individual with the derived allele (supported by two reads) is the ANE individual Afontova Gora 3, which is directly dated to 16130-15749 cal BCE (14710±60 BP, MAMS-27186: a previously unpublished date that we newly report here). We cannot determine the status of rs12821256 in Afontova Gora 2 and MA-1 due to lack of sequence coverage at this SNP." www.biorxiv.org/content/biorxiv/suppl/2017/09/19/135616.DC4/135616-1.pdf |
|
|
|
Post by Admin on Aug 13, 2023 9:20:35 GMT
Most Europeans lacked two depigmentation genes, SLC24A5 and SLC45A2, about 8,500 years ago. But the 7,700-year-old Motala archaeological site in southern Sweden had both light skin gene variants, SLC24A5 and SLC45A2. Ancient hunter-gatherers (SHGs) appeared in Scandinavia around 11,000 BP without SLC24A5 and SLC45A2, which are associated with skin pigmentation, while they were already blue-eyed with HERC2/OCA2. SHGs in the Motala archaeological site in southern Sweden became pale by 7,700 years ago through admixing with eastern hunter-gatherers (EHGs) from today's Ukraine or Russia.  Population genomics of Mesolithic Scandinavia: Investigating early postglacial migration routes and high-latitude adaptation In addition to performing this genome-wide scan, we studied the allele frequencies in three pigmentation genes (SLC24A5, SLC45A2, which have a strong effect on skin pigmentation, and OCA2/HERC2, which has a strong effect on eye pigmentation) in which the derived alleles are virtually fixed in northern Europeans today. The differences in allele frequencies of those three loci are among the highest between human populations, suggesting that selection was driving the differences in eye color, skin, and hair pigmentation as part of the adaptation to different environments [50–53]. All of the depigmentation variants at these three genes are in high frequency in SHGs in contrast to both WHGs and EHGs (Fig 4B). We conduct neutral simulations of the allele frequencies in an admixed SHG population to estimate p-values for observing these allele frequencies without selection (S9 Text). The p-values for all three SNPs are lower than 0.2; the combined p-value [54] for all three pigmentation SNPs is 0.028. Therefore, the unique configuration of the SHGs is not fully explained by the fact that SHGs are a mixture of EHGs and WHGs, but could rather be explained by a continued increase of the allele frequencies after the admixture event, likely caused by adaptation to high-latitude environments [50,52]. journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.2003703Genetic continuity, isolation, and gene flow in Stone Age Central and Eastern Europe Abstract The genomic landscape of Stone Age Europe was shaped by multiple migratory waves and population replacements, but different regions do not all show similar patterns. To refine our understanding of the population dynamics before and after the dawn of the Neolithic, we generated and analyzed genomic sequence data from human remains of 56 individuals from the Mesolithic, Neolithic, and Eneolithic across Central and Eastern Europe. We found that Mesolithic European populations formed a geographically widespread isolation-by-distance zone ranging from Central Europe to Siberia, which was already established 10,000 years ago. We found contrasting patterns of population continuity during the Neolithic transition: people around the lower Dnipro Valley region, Ukraine, showed continuity over 4000 years, from the Mesolithic to the end of the Neolithic, in contrast to almost all other parts of Europe where population turnover drove this cultural change, including vast areas of Central Europe and around the Danube River. www.nature.com/articles/s42003-023-05131-3 |
|
|
|
Post by Admin on Aug 14, 2023 2:35:34 GMT
Population genomics of Mesolithic Scandinavia: Investigating early postglacial migration routes and high-latitude adaptation Abstract Scandinavia was one of the last geographic areas in Europe to become habitable for humans after the Last Glacial Maximum (LGM). However, the routes and genetic composition of these postglacial migrants remain unclear. We sequenced the genomes, up to 57× coverage, of seven hunter-gatherers excavated across Scandinavia and dated from 9,500–6,000 years before present (BP). Surprisingly, among the Scandinavian Mesolithic individuals, the genetic data display an east–west genetic gradient that opposes the pattern seen in other parts of Mesolithic Europe. Our results suggest two different early postglacial migrations into Scandinavia: initially from the south, and later, from the northeast. The latter followed the ice-free Norwegian north Atlantic coast, along which novel and advanced pressure-blade stone-tool techniques may have spread. These two groups met and mixed in Scandinavia, creating a genetically diverse population, which shows patterns of genetic adaptation to high latitude environments. These potential adaptations include high frequencies of low pigmentation variants and a gene region associated with physical performance, which shows strong continuity into modern-day northern Europeans. Author summary The Scandinavian peninsula was the last part of Europe to be colonized after the Last Glacial Maximum. The migration routes, cultural networks, and the genetic makeup of the first Scandinavians remain elusive and several hypotheses exist based on archaeology, climate modeling, and genetics. By analyzing the genomes of early Scandinavian hunter-gatherers, we show that their migrations followed two routes: one from the south and another from the northeast along the ice-free Norwegian Atlantic coast. These groups met and mixed in Scandinavia, creating a population more diverse than contemporaneous central and western European hunter-gatherers. As northern Europe is associated with cold and low light conditions, we investigated genomic patterns of adaptation to these conditions and genes known to be involved in skin pigmentation. We demonstrate that Mesolithic Scandinavians had higher levels of light pigmentation variants compared to the respective source populations of the migrations, suggesting adaptation to low light levels and a surprising signal of genetic continuity in TMEM131, a gene that may be involved in long-term adaptation to the cold.  Fig 1. Mesolithic samples and their genetic affinities. (A) Map of the Mesolithic European samples used in this study. The pie charts show the model-based [18,19] estimates of genetic ancestry for each SHG individual. The map also displays the ice sheet covering Scandinavia 10,000 cal BP (most credible [solid line] and maximum extend [dashed line] following [10]). Newly sequenced individuals are shown with bold and italic site names. SF11 is excluded from this map due to its low coverage (0.1×). Additional European EHG and WHG individuals used in this study derive from sites outside this map. The map was plotted using the R package rworldmap [28]. (B) Magnified section of genetic similarity among ancient and modern day individuals using PCA, featuring only the Mesolithic European samples (see S6 Text for the full plot). Symbols representing newly sequenced individuals have a black contour line. (C) Allele sharing between the SHGs, Latvian Mesolithic hunter-gatherers (Zv) [29], and EHGs versus WHGs measured by the statistic f4(Chimp, SHG; EHG, WHG) calculated for the captured SNPs [20]. Error bars show two block-jackknife standard errors. Data shown in this figure can be found in S1 Data. BP, before present; cal, calibrated; Chimp, Chimpanzee; EHG, eastern hunter-gatherer; PCA, principal component analysis; SHG, Scandinavian hunter-gatherer; WHG, western hunter-gatherer; Zv, Latvian Mesolithic hunter-gatherer from Zvejnieki. doi.org/10.1371/journal.pbio.2003703.g001Introduction As the ice sheet retracted from northern Europe after the Last Glacial Maximum (LGM), around 23,000 years ago, new habitable areas emerged [1], allowing plants [2,3] and animals [4,5] to recolonize the Scandinavian peninsula (hereafter referred to as Scandinavia). There is consistent evidence of human presence in the archaeological record from approximately 11,700 years before present (BP) both in southern and northern Scandinavia [6–9]. At this time, the ice sheet was still dominating the interior of Scandinavia [9,10] (Fig 1A, S1 Text), but recent climate modeling shows that the Arctic coast of (modern-day) northern Norway was ice free [10]. Similarities in late-glacial lithic technology (direct blade percussion technique) of Western Europe and the oldest counterparts of Scandinavia appearing around 11,000 calibrated (cal) BP [11] (S1 Text) have been used to argue for an early postglacial migration from southwestern Europe into Scandinavia, including areas of northern Norway. However, studies of another lithic technology, the “pressure blade” technique, which first occurred in the northern parts of Scandinavia around 10,200 cal BP, indicates contact with groups in the east and possibly an eastern origin of the early settlers [7,12–15] (S1 Text). The first genetic studies of Mesolithic human remains from central and eastern Scandinavian hunter-gatherers (SHGs) revealed similarities to two different Mesolithic European populations, the “western hunter-gatherers” (WHGs) from western, central, and southern Europe and the “eastern hunter-gatherers” (EHGs) from northeastern and eastern Europe [16–24]. Archaeology, climate modeling, and genetics suggest several possibilities for the early postglacial migrations into Scandinavia, including migrations from the south, southeast, northeast, and combinations of these; however, the early postglacial peopling of Scandinavia remains elusive [1,4,6–19,25,26]. In this study, we contrast genome sequence data and stable isotopes from Mesolithic human remains from western, northern, and eastern Scandinavia to infer the early postglacial migration routes into Scandinavia—from where people came, what routes they followed, how they were related to other Mesolithic Europeans [17–21,27]—and to investigate human adaptation to high-latitude environments. |
|
|
|
Post by Admin on Aug 14, 2023 23:21:28 GMT
Results and discussion We sequenced the genomes of seven hunter-gatherers from Scandinavia (Table 1; S1, S2 and S3 Text) ranging from 57.8× to 0.1× genome coverage, of which four individuals had a genome coverage above 1×. The remains were directly dated to between 9,500 cal BP and 6,000 cal BP, and were excavated in southwestern Norway (Hum1, Hum2), northern Norway (Steigen), and the Baltic islands of Stora Karlsö and Gotland (SF9, SF11, SF12, and SBj), and represent 18% (6 of 33) of all known human remains in Scandinavia older than 8,000 years [30]. All samples displayed fragmentation and cytosine deamination at fragment termini characteristic for ancient DNA (aDNA) (S3 Text). Mitochondrial (mt) DNA-based contamination estimates were <6% for all individuals (confidence intervals ranging from 0% to 9.5%) and autosomal contamination was <1% for all individuals except for SF11, which showed approximately 10% contamination (Table 1, S4 Text). Four of the seven individuals were inferred to be males and three were females. All the western and northern Scandinavian individuals and one eastern Scandinavian carried U5a1 mt haplotypes, whereas the remaining eastern Scandinavians carried U4a haplotypes (Table 1, S5 Text). These individuals represent the oldest U5a1 and U4 lineages detected so far. The Y chromosomal haplotype was determined for three of the four males, all carried I2 haplotypes, which were common in pre-Neolithic Europe (Table 1, S5 Text). 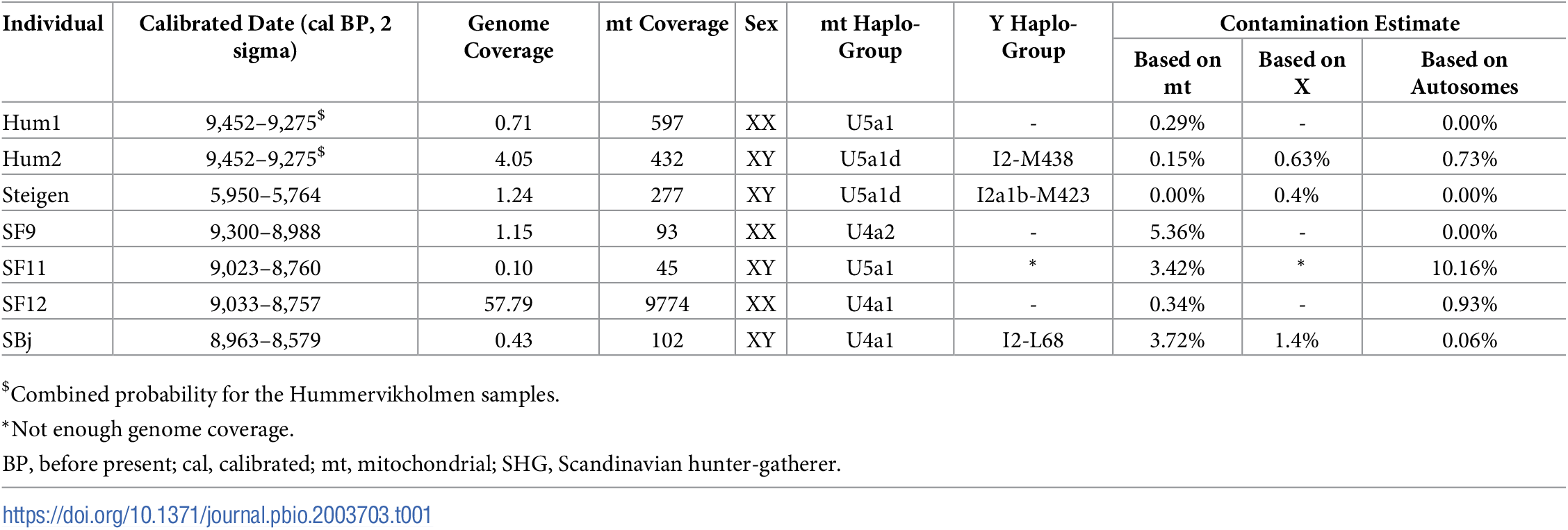 Table 1. Information on the seven SHGs investigated in this study, including cal BP (corrected for the marine reservoir effect, given as a range of two standard deviations), average genome coverage, average mt coverage, mt and Y chromosome haplogroups, and contamination estimates based on the mt, the X-chromosome for males and the autosomes. doi.org/10.1371/journal.pbio.2003703.t001The high coverage and Uracil-DNA-glycosylase (UDG)-treated genome (used in order to reduce the effects of postmortem DNA damage) [31] of SF12 allowed us to confidently discover new and hitherto unknown variants at sites with 55× or higher sequencing depth (S3 Text). Based on SF12’s high-coverage and high-quality genome, we estimate the number of SNPs hitherto unknown (not recorded in dbSNP [v142]) to be approximately 10,600. This number is close to the median per European individual in the 1000 Genomes Project [32] (approximately 11,400, S3 Text), although a direct comparison is difficult due to the lower sequencing depth, different data processing, and larger sample sizes in the 1000 Genomes Project. At least 17% of these SNPs that are not found in modern-day individuals were in fact common among the Mesolithic Scandinavians (seen in the low coverage data conditional on the observation in SF12), and in total 24.2% were found in other prehistoric individuals (S3 Text), suggesting a substantial amount of hitherto unknown variation 9,000 years ago (S3 Text). Thus, many genetic variants found in Mesolithic individuals have not been carried over to modern-day groups. Among the novel variants in SF12, four (all heterozygous) are predicted to affect the function of protein coding genes [33] (S3 Text). The “heat shock protein” HSPA2 in SF12 carries an unknown mutation that changes the amino acid histidine to tyrosine at a protein–protein interaction site, which likely disrupts the function of the protein (S3 Text). Defects in HSPA2 are known to drastically reduce fertility in males [34]. It will be interesting to see how common such variants were among Mesolithic groups as more genome sequence data become available. The genomic data further allowed us to study the physical appearance of SHGs (S8 Text); for instance, they show a combination of eye color varying from blue to light brown and light skin pigmentation. This is strikingly different from the WHGs—who have been suggested to have the specific combination of blue eyes and dark skin [18,20,21,23] and EHGs—who have been suggested to be brown-eyed and light-skinned [19,20]. |
|





