Post by Admin on Nov 20, 2023 21:20:54 GMT
Multi-ancestry genome-wide association study of cannabis use disorder yields insight into disease biology and public health implications
Abstract
As recreational use of cannabis is being decriminalized in many places and medical use widely sanctioned, there are growing concerns about increases in cannabis use disorder (CanUD), which is associated with numerous medical comorbidities. Here we performed a genome-wide association study of CanUD in the Million Veteran Program (MVP), followed by meta-analysis in 1,054,365 individuals (ncases = 64,314) from four broad ancestries designated by the reference panel used for assignment (European n = 886,025, African n = 123,208, admixed American n = 38,289 and East Asian n = 6,843). Population-specific methods were applied to calculate single nucleotide polymorphism-based heritability within each ancestry. Statistically significant single nucleotide polymorphism-based heritability for CanUD was observed in all but the smallest population (East Asian). We discovered genome-wide significant loci unique to each ancestry: 22 in European, 2 each in African and East Asian, and 1 in admixed American ancestries. A genetically informed causal relationship analysis indicated a possible effect of genetic liability for CanUD on lung cancer risk, suggesting potential unanticipated future medical and psychiatric public health consequences that require further study to disentangle from other known risk factors such as cigarette smoking.
Main
Cannabis is a psychoactive substance with a long history of use and dependence. Recently within the United States, 37 states have approved what is termed medical cannabis use, and 19 states, 2 territories and the District of Columbia allow possession of cannabis for recreational purposes. In Europe, only Malta has fully legalized recreational cannabis, although many other countries have decriminalized possession of small amounts of cannabis and have enabled medical allowances. It was recently legalized in Thailand but remains prohibited in many parts of Asia, the Middle East and South America. The status in many of these places may be subject to change in the near future. More than a third of individuals who use cannabis develop cannabis use disorders (CanUD), and evidence regarding the impact of legalization on escalating use and use disorders is mixed1,2. Substantial negative health outcomes associated with chronic cannabis use include various cancers associated with inhaling combustion products3, declines in cognitive capacity and motivation and increased schizophrenia (SCZ) risk4,5. Individual and societal complications that result from CanUD include decreased productivity and accidents related to intoxication6. The full range of risks and negative outcomes associated with cannabis use and CanUD may not be appreciated widely. Considering the gradually increasing permissiveness surrounding its use, understanding various sources of risk that influence CanUD is both necessary and timely.
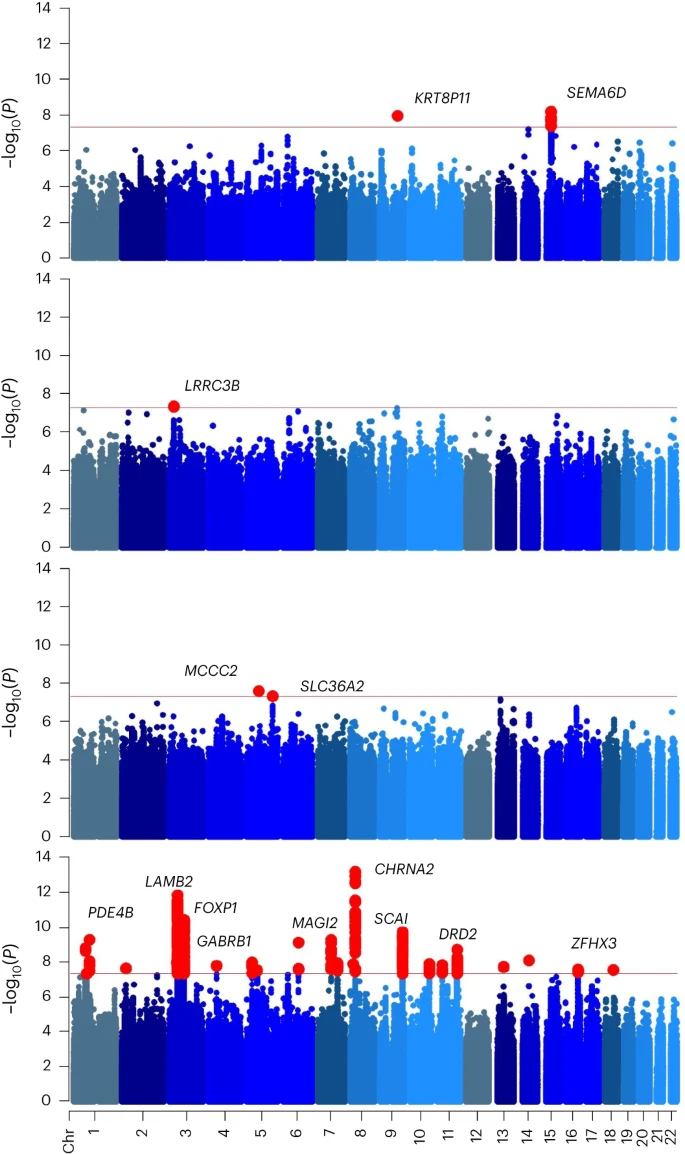
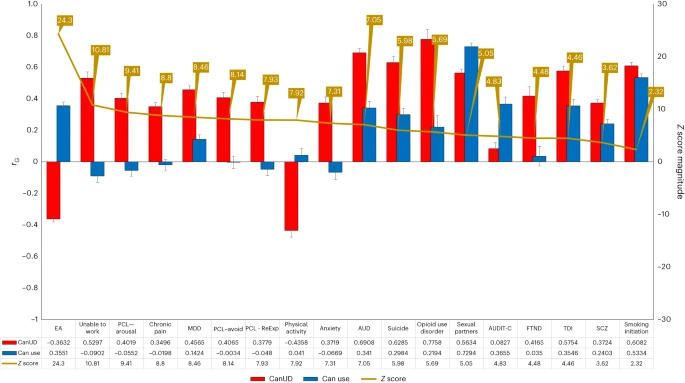
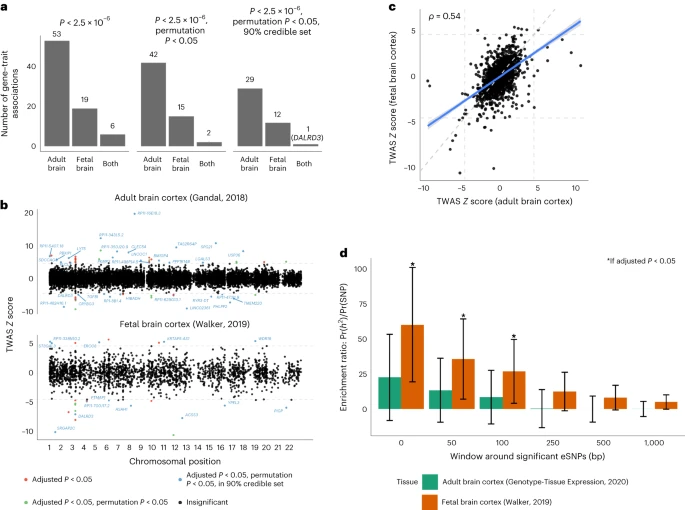
In this Article, we combined genome-wide genotype data from the Million Veteran Program (MVP) with expanded samples from iPSYCH27,8 and Mass General Brigham (MGB) BioBank9 and meta-analyzed these with the Psychiatric Genomics Consortium (PGC)/deCODE/iPSYCH1 study7,10. MVP, one of the largest biobanks in the world11, has enabled a substantial increase in power for genomic discovery by doubling the number of cases of European (EUR) ancestry available. By increasing sample numbers, we substantially increased the number of discovered loci and confirmed previous findings7,10. We also leveraged the ancestral diversity of the MVP to expand analyses of African ancestry individuals (AFR) and conducted genome-wide association studies (GWAS) analyses in Admixed American (AMR) and East Asian (EAS) ancestries. Linkage disequilibrium (LD) score regression (LDSC) can quantify variance explained by genetics and identify overlap between traits. This method is sufficient for EUR ancestries but not appropriate for some non-European and admixed ancestries. To solve this problem, we used cohort-derived covariate LDSC12 to calculate single nucleotide polymorphism (SNP)-based heritability in these populations, finding similar results among all ancestries. We conducted a transcriptome-wide association study (TWAS), which leverages annotations based on variant associations to changes in gene expression, in adult and fetal brain tissue to identify significant expression quantitative trait loci (eQTLs), using stratified LDSC to show enriched SNP-based heritability in fetal but not adult cortex. We also conducted Mendelian randomization (MR) analyses—an approach that uses genetic variations identified by GWAS as instruments to obtain an unbiased estimate of the effect of a trait of interest (here, CanUD) on outcomes—to examine causal relationships with chronic pain, lung cancer, physical activity and SCZ. Finally, we performed genomic structural equation modeling (gSEM)—a multivariate method for analyzing GWAS summary statistics to examine joint genetic architecture of traits—to understand the genomic relationships between cannabis use traits and other psychiatric and substance use disorder (SUD) traits. This work builds upon a decade of progress in the field7,10,13,14,15,16,17,18.
www.nature.com/articles/s41588-023-01563-z
Abstract
As recreational use of cannabis is being decriminalized in many places and medical use widely sanctioned, there are growing concerns about increases in cannabis use disorder (CanUD), which is associated with numerous medical comorbidities. Here we performed a genome-wide association study of CanUD in the Million Veteran Program (MVP), followed by meta-analysis in 1,054,365 individuals (ncases = 64,314) from four broad ancestries designated by the reference panel used for assignment (European n = 886,025, African n = 123,208, admixed American n = 38,289 and East Asian n = 6,843). Population-specific methods were applied to calculate single nucleotide polymorphism-based heritability within each ancestry. Statistically significant single nucleotide polymorphism-based heritability for CanUD was observed in all but the smallest population (East Asian). We discovered genome-wide significant loci unique to each ancestry: 22 in European, 2 each in African and East Asian, and 1 in admixed American ancestries. A genetically informed causal relationship analysis indicated a possible effect of genetic liability for CanUD on lung cancer risk, suggesting potential unanticipated future medical and psychiatric public health consequences that require further study to disentangle from other known risk factors such as cigarette smoking.
Main
Cannabis is a psychoactive substance with a long history of use and dependence. Recently within the United States, 37 states have approved what is termed medical cannabis use, and 19 states, 2 territories and the District of Columbia allow possession of cannabis for recreational purposes. In Europe, only Malta has fully legalized recreational cannabis, although many other countries have decriminalized possession of small amounts of cannabis and have enabled medical allowances. It was recently legalized in Thailand but remains prohibited in many parts of Asia, the Middle East and South America. The status in many of these places may be subject to change in the near future. More than a third of individuals who use cannabis develop cannabis use disorders (CanUD), and evidence regarding the impact of legalization on escalating use and use disorders is mixed1,2. Substantial negative health outcomes associated with chronic cannabis use include various cancers associated with inhaling combustion products3, declines in cognitive capacity and motivation and increased schizophrenia (SCZ) risk4,5. Individual and societal complications that result from CanUD include decreased productivity and accidents related to intoxication6. The full range of risks and negative outcomes associated with cannabis use and CanUD may not be appreciated widely. Considering the gradually increasing permissiveness surrounding its use, understanding various sources of risk that influence CanUD is both necessary and timely.
In this Article, we combined genome-wide genotype data from the Million Veteran Program (MVP) with expanded samples from iPSYCH27,8 and Mass General Brigham (MGB) BioBank9 and meta-analyzed these with the Psychiatric Genomics Consortium (PGC)/deCODE/iPSYCH1 study7,10. MVP, one of the largest biobanks in the world11, has enabled a substantial increase in power for genomic discovery by doubling the number of cases of European (EUR) ancestry available. By increasing sample numbers, we substantially increased the number of discovered loci and confirmed previous findings7,10. We also leveraged the ancestral diversity of the MVP to expand analyses of African ancestry individuals (AFR) and conducted genome-wide association studies (GWAS) analyses in Admixed American (AMR) and East Asian (EAS) ancestries. Linkage disequilibrium (LD) score regression (LDSC) can quantify variance explained by genetics and identify overlap between traits. This method is sufficient for EUR ancestries but not appropriate for some non-European and admixed ancestries. To solve this problem, we used cohort-derived covariate LDSC12 to calculate single nucleotide polymorphism (SNP)-based heritability in these populations, finding similar results among all ancestries. We conducted a transcriptome-wide association study (TWAS), which leverages annotations based on variant associations to changes in gene expression, in adult and fetal brain tissue to identify significant expression quantitative trait loci (eQTLs), using stratified LDSC to show enriched SNP-based heritability in fetal but not adult cortex. We also conducted Mendelian randomization (MR) analyses—an approach that uses genetic variations identified by GWAS as instruments to obtain an unbiased estimate of the effect of a trait of interest (here, CanUD) on outcomes—to examine causal relationships with chronic pain, lung cancer, physical activity and SCZ. Finally, we performed genomic structural equation modeling (gSEM)—a multivariate method for analyzing GWAS summary statistics to examine joint genetic architecture of traits—to understand the genomic relationships between cannabis use traits and other psychiatric and substance use disorder (SUD) traits. This work builds upon a decade of progress in the field7,10,13,14,15,16,17,18.
www.nature.com/articles/s41588-023-01563-z