Post by Admin on Dec 8, 2023 19:01:10 GMT
Fig. 4: gSEM.
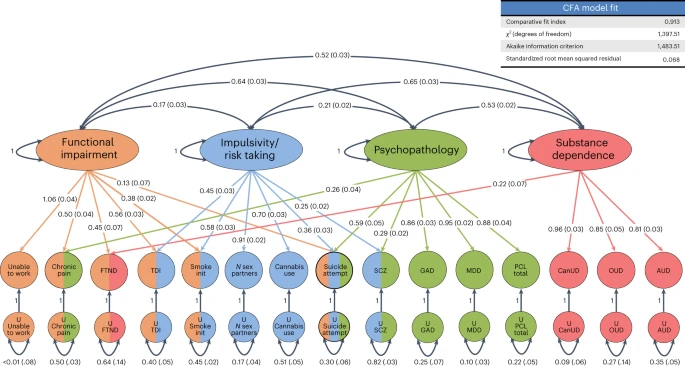
gSEM was used to cluster 14 traits correlated with CanUD. Exploratory and confirmatory factor analysis indicated four factors fit the data best. Factors fit mostly into categories that we designated as functional impairment (factor 1), impulsivity and risk taking (factor 2), psychopathology (factor 3) and substance dependence (factor 4). CanUD fit best (and strongly) in the substance dependence cluster. FTND fit into factors for functional impairment and substance dependence. Suicide attempts fit into functional impairment, impulsivity and risk taking, and psychopathology. Numbers outside the parenthesis are correlation coefficients between factors. Numbers inside parenthesis are stadard errors of means. Smoke init, initiation of regular smoking; GAD, generalized anxiety disorder; OUD, opioid use disorder.
Full size image
Discussion
Recently, cannabis use has been legalized in various US states and elsewhere without fully examining the health consequences of individual or societal risks. An epidemiologic survey conducted by the National Survey on Drug Use and Health in the United States identified a past-year cannabis use prevalence of 17.5%, an increase from 11.0% in 2002, and 1.8% with CanUD, the same percentage recorded in 2002. Usage varies worldwide, with many regions of high prevalence21.
The findings we report here add to our understanding of CanUD biology on many levels. First, we greatly increased the available sample size for genomic analysis, mostly by incorporating MVP data, and identified multiple novel risk loci in four populations, improving on previous results in EUR by more than an order of magnitude and presenting the first genetic discoveries in the other populations studied. Using the GWAS data, we then showed overlapping genetic liability to other traits. Next, investigating how genetic variation underlying CanUD influences fetal brain gene expression, the brain in particular showed significant enrichment for SNP-based heritability. Essentially, SNPs that influence fetal brain gene expression explain a greater proportion of CanUD phenotypic variance than the overall GWAS association of all SNPs. We investigated the overlapping and shared underlying genetic architectures of several different traits and employed MR to demonstrate putative causal relationships between outcomes with substantial impact on human health, including an association with lung cancer risk. Cannabis is frequently consumed using methods involving inhaling combustion products, potentially exposing users to risks similar to those found in smoking other substances such as tobacco. Indeed, some of the shared genetic risk between CanUD and tobacco smoking may relate to propensity to smoke per se, independent of substance, a hypothesis that we currently lack the power to evaluate.
We identified 22 significant loci, most of them novel, for CanUD in EUR. We also replicated findings in CHRNA2 (meta P = 7.3 × 10−14, MVP only P = 1.1 × 10−5) and FOXP2 (meta P = 1.7 × 10−8, MVP only P = 2.0 × 10−3), with triple the effective sample size of the largest of those studies10, demonstrating once again the stability of GWAS findings as sufficient sample size and power to discover new loci are reached22,23. We discovered GWS loci in four ancestral groups: EUR, AFR, AMR and EAS. In AFR, two independent SNPs were associated on chromosome 5. The first (rs574008891) was within an intron of the gene that encodes methylcrotonyl-CoA carboxylase subunit 2 (MCCC2). The other significant locus (rs573117193) mapped to an intron in the solute carrier family 36 member 2 (SLC36A2) gene. These specific variants are absent in the other ancestries studied. For AMR, the one risk locus was rare (rs9815757, minor allele frequency (MAF) 0.1%) and mapped in an intergenic region downstream of leucine rich repeat containing 3B (LRRC3B). Finally, for EAS, one locus was associated with CanUD: rs78561048, near semaphorin 6D (SEMA6D). Follow-up analysis in larger samples is needed to assess the robustness of findings, particularly in AMR and EAS. Several variants showed concordant direction of effect across all four stratified ancestral groups (Table 1). For instance, rs10986600, significantly associated in EUR on chromosome 9, was nominally significant (P < 0.05) with same effect direction in AFR (0.04) and AMR (0.03) and significant in the multi-ancestry meta-analysis. This intronic variant of the protein phosphatase 6 catalytic subunit (PPP6C) is an eQTL for PPP6C, a gene linked to various cancers, including skin melanoma and lung squamous cell carcinoma. Multi-ancestry meta-analysis revealed an additional five loci not identified in the stratified analyses. Among them, the lead SNP on chromosome 15, rs147144681, which maps to an intron of the cholinergic receptor nicotinic alpha 3 subunit (CHRNA3) gene, is particularly noteworthy; as reported above, variation in CHRNA2 was among the first variants associated with CanUD and was replicated here. This suggests potential convergence involving the cholinergic system broadly and nicotinic receptors, specifically in the underlying etiology of CanUD. While nicotinic receptors are also associated with tobacco smoking-related traits24, the relative pattern of association for those traits is different from the observations for CanUD—for many smoking-related traits, a chromosome 15 nicotinic receptor cluster is associated with orders of magnitude greater support than other variants, including other nicotinic receptors; for CanUD, CHRNA2 is consistently the strongest association, also by orders of magnitude. We conducted conditional analysis for CHRNA2 and found the conditional P value remained robust following conditioning on smoking initiation20 (Pcond = 4.6 × 10−14). This replicates similar analyses performed by Demontis et al.7 and Johnson et al.10, which showed conditioning on smoking did not affect the CanUD association at this variant. Several other loci near cholinergic receptor subunit genes previously identified for smoking are not significant in our analysis of CanUD (CHRNA4, rs13036436, smoking P = 1.1 × 10−29, CanUD P = 0.97; CHRNA5, rs667282, smoking P = 9.9 × 10−25, CanUD P = 0.043). Conversely, the CHRNA3 variant we find associated with CanUD is not significant for smoking (rs147144681, smoking P = 0.0033, CanUD P = 3.3 × 10−8) (ref. 20).
Genetic correlations were calculated for 1,335 traits to identify genetic overlap with CanUD. Some traits with significant rG were tested for causal inference based on a combination of significant genetic correlation and a prior interest in phenotype (physical activity, multi-site chronic pain, Alzheimer’s disease and SCZ). We identified a bidirectional causal relationship between CanUD and SCZ. At the same time, the MR Egger analysis indicated this was not due to horizontal pleiotropy. This supports similar findings reported previously, confirming previous genetic–epidemiologic studies25 and verifying an important public health risk associated with CanUD. To highlight differences between cannabis use and CanUD, we compared the pattern of genetic correlations across 18 traits, which showed striking differences. CanUD was much more closely associated with psychopathology, recapitulating a general pattern seen with other comparisons of SUD and use traits26. For example, while we observed a substantial negative correlation between CanUD and educational attainment, cannabis use was associated with greater educational attainment. POPCORN was used to generate a cross-covariance score to allow for comparison of traits across ancestries using genetic correlations for EUR and AFR groups (Supplementary Fig. 3). We found a striking similarity for cross trait comparisons for both groups, indicating a similar underlying genomic architecture. This finding supports the possibility that some findings uncovered so far for EUR individuals, recruited in vastly greater numbers for genetic study, will provide some degree of generalizability across human populations.
Chronic pain may be a factor driving CanUD in some individuals, with significant unidirectional evidence for a causal effect of chronic pain27 on CanUD in the MR analysis. Cannabis use has been proposed as a treatment for chronic pain, and there are several clinical trials in progress28. This MR observation suggests that there may be merit in cannabis as a treatment for at least some kinds of pain. The small overall beneficial effect observed requires so many individuals to be treated that harmful effects (such as increased CanUD) also become a significant factor29. Our MR results suggesting that chronic pain has a causal influence on CanUD emphasize the need for follow-up investigations that address whether greater consideration should be given to the adverse effects, rather than just the therapeutic effects among individuals receiving cannabis-based medicines. A similar question arises with opioids, which although often prescribed for pain, can also cause great harm30: namely, what level of risk of CanUD is acceptable given cannabis’ potential to improve quality of life and reduce opioid exposure in chronic pain patients? Our results suggest that harms such as dependence and consequences, reflected in underlying genetics of the trait, may need to be weighed against the potential benefits of cannabis treatment for chronic pain. Future studies should consider this novel relationship to pain31 and clinical efficacy trials are underway.
Cigarette smoking substantially increases the risk of many forms of cancer, including lung cancer, through numerous well-studied mechanisms with established literature dating back more than 60 years32. The influence of cannabis on cancer risk is less well understood; it should be anticipated that these combustion products could have harmful pulmonary impacts—indeed, it would be surprising if smoking tobacco, but not smoking cannabis, increased cancer risk. MR yielded evidence for a unidirectional causal effect of CanUD on lung cancer. This result was robust to conditioning on data from the largest available smoking initiation GWAS but not conditioning on cigarettes per day, both traits that also have causal relationships with lung cancer but far more robust genetic instruments to evaluate this relationship. We do not currently have a way to assess genetic variation associated with the route of cannabis administration, but combustion is by far the most common method in the MVP and other cohorts studied. Given the trend toward increased legalization and usage, this apparent causal association needs to be monitored as it may have profound and underappreciated public health consequences. As the causal relationship with CanUD was not robust to conditioning on cigarettes per day, one probable explanation may be that there is horizontal pleiotropy between these traits in their influence on lung cancer.
Four GWS loci overlapped with TWAS prioritization from the EUR meta-analysis, using eQTL integration from samples of adult33 and fetal34 cortical tissue. These were DALRD3 (both fetal and adult), ERCC8 (fetal), RP11-629G13.1 (adult) and PHLPP2 (adult). The DALRD3 protein product, a DALR anticodon binding domain, forms a complex with the product of METTL2B. Nonsense mutations in DALRD3 are associated with developmental delay and early-onset epileptic encephalopathy35. ERCC8 encodes the excision repair 8, CSA ubiquitin complex subunit, which plays a role in DNA repair and is associated with the developmental disorder Cockayne syndrome36, as well as breast, esophageal and other cancers37,38. RP11-629G13.1 is a long noncoding RNA associated with downregulation of NCAM1 gene expression in multiple myeloma patients39. Significant partitioned SNP-based heritability was observed in fetal but not in the adult cortex, with 4.36% of trait SNP-based heritability explained by 0.12% of the total SNPs near fetal frontal cortex eQTLs. Only 1.77% of CanUD SNP-based heritability was explained using 0.13% of the total SNPs near adult cortex eQTLs. Fetal development may play a role in SUD susceptibility40, and substance use can influence fetal development during pregnancy and health outcomes during childhood41. Although exogenous exposure to cannabis may not occur until years or decades after birth, enriched fetal SNP-based heritability in this study argues a possible role for genetic effects on CanUD in the developing brain independent of exposure. SCZ risk is also modulated by risk factors during fetal development42 and genetic43 and environmental effects (including maternal food deprivation in the first trimester of pregnancy44). Temporal convergence of the initiation of genetic risk effects for both SCZ and CanUD, if validated experimentally, would provide insight into the genetic relationship between these disorders and could relate to a mechanism for the bidirectional risk relationship between cannabis use and SCZ.
gSEM was used to contextualize summary statistics from this project with those from other published GWAS studies. Exploratory and confirmatory factor analyses showed that four factors provide the best fit for the 14 correlated traits included in the analysis. Factors fit mostly into categories that relate to functional impairment (factor 1), impulsivity and risk taking (factor 2), psychopathology (factor 3) and substance dependence (factor 4). CanUD fit best (and strongly) in the substance dependence cluster (factor 4). FTND fit into factors for functional impairment and substance dependence. Suicide attempts fit into functional impairment, impulsivity/risk taking and psychopathology. This is consistent with research showing overlapping pathologies within addiction and shared genetic risk factors between them45.
This study has limitations. The use of electronic health records allows for a large sample of CanUD cases but limits the assessment of subdiagnostic cannabis use in controls. Although we accounted for subdiagnostic cannabis users by excluding them from controls when information was available, these are probably underreported. Future studies of individuals with ascertained cannabis use who do not meet criteria for CanUD would provide more insight into the specific genetic liability to dependence. As the traits of interest were gathered from previously published reports or queries of electronic health records (EHRs) for diagnostic codes, we did not have information regarding tetrahydrocannabinol (THC) blood levels or information on the potency of cannabis at each exposure. If these data were available, study of effects on cannabis potency on dependence and comorbidities would be of great interest. We identified a causal relationship between multi-site chronic pain and CanUD. As pain is a complex trait and different type of pain may interact differently with CanUD, our finding for multi-site chronic pain is not sufficient to draw conclusions about the interaction between CanUD and specific kinds of pain or pain syndromes. Our definition of CanUD was based on any report of abuse or dependence either as an inpatient or outpatient. Participants in this study span a period of changing legal status and increasing use of marijuana, a major secular trend. Given the age of the participants (Supplementary Table 16) and expected time from initial exposure to the development of a use disorder, nearly all participants would have been exposed to cannabis before legalization. The TWAS study did not include ascertainment for CanUD in the individuals who donated brain tissue used for analysis. We discovered GWS loci in ancestral groups, but AFR, AMR and EAS sample sizes were small compared to EUR. We did not perform MR or TWAS analyses in non-European samples because available GWAS and eQTL datasets are still limited in non-European ancestry populations, and cross-ancestry analyses carry risk of biases due to differences in the underlying LD structure between ancestries. More studies are needed of individuals of diverse ancestries to replicate these findings, estimate their robustness and ensure that the benefits provided by these studies are available to all people.
This is the largest genetic study of CanUD so far, including data from multiple international cohorts in more than one million participants and comprising four ancestral groups. We replicate two prior GWS findings while identifying 25 novel loci, and we leverage these novel data to investigate genetic overlap with other traits. We identify a clear difference between cannabis use and CanUD, with genetic liability to CanUD being much more closely associated with psychopathology and disability. We found greater heritability enrichment in fetal than adult brain tissue, supporting an important role of development in laying the biological basis for CanUD. We used MR to assess causal relationships and found evidence of bidirectional causal effects between CanUD and SCZ and unidirectional effects of multi-site chronic pain on CanUD, and of CanUD on lung cancer. Finally, using gSEM, we found that CanUD loads on a latent factor with other substance dependence traits, consistent with clinical observation, genetic epidemiology and prior genetic studies of other SUD traits. In particular, we highlight the possible relationship revealed herein between CanUD and lung cancer risk. This study yields new insights into the genetic architecture of CanUD and how this risk interacts with traits crucial to public health and raises important concerns regarding the potential adverse consequences of the secular trend toward increased cannabis use consequent to legalization.

gSEM was used to cluster 14 traits correlated with CanUD. Exploratory and confirmatory factor analysis indicated four factors fit the data best. Factors fit mostly into categories that we designated as functional impairment (factor 1), impulsivity and risk taking (factor 2), psychopathology (factor 3) and substance dependence (factor 4). CanUD fit best (and strongly) in the substance dependence cluster. FTND fit into factors for functional impairment and substance dependence. Suicide attempts fit into functional impairment, impulsivity and risk taking, and psychopathology. Numbers outside the parenthesis are correlation coefficients between factors. Numbers inside parenthesis are stadard errors of means. Smoke init, initiation of regular smoking; GAD, generalized anxiety disorder; OUD, opioid use disorder.
Full size image
Discussion
Recently, cannabis use has been legalized in various US states and elsewhere without fully examining the health consequences of individual or societal risks. An epidemiologic survey conducted by the National Survey on Drug Use and Health in the United States identified a past-year cannabis use prevalence of 17.5%, an increase from 11.0% in 2002, and 1.8% with CanUD, the same percentage recorded in 2002. Usage varies worldwide, with many regions of high prevalence21.
The findings we report here add to our understanding of CanUD biology on many levels. First, we greatly increased the available sample size for genomic analysis, mostly by incorporating MVP data, and identified multiple novel risk loci in four populations, improving on previous results in EUR by more than an order of magnitude and presenting the first genetic discoveries in the other populations studied. Using the GWAS data, we then showed overlapping genetic liability to other traits. Next, investigating how genetic variation underlying CanUD influences fetal brain gene expression, the brain in particular showed significant enrichment for SNP-based heritability. Essentially, SNPs that influence fetal brain gene expression explain a greater proportion of CanUD phenotypic variance than the overall GWAS association of all SNPs. We investigated the overlapping and shared underlying genetic architectures of several different traits and employed MR to demonstrate putative causal relationships between outcomes with substantial impact on human health, including an association with lung cancer risk. Cannabis is frequently consumed using methods involving inhaling combustion products, potentially exposing users to risks similar to those found in smoking other substances such as tobacco. Indeed, some of the shared genetic risk between CanUD and tobacco smoking may relate to propensity to smoke per se, independent of substance, a hypothesis that we currently lack the power to evaluate.
We identified 22 significant loci, most of them novel, for CanUD in EUR. We also replicated findings in CHRNA2 (meta P = 7.3 × 10−14, MVP only P = 1.1 × 10−5) and FOXP2 (meta P = 1.7 × 10−8, MVP only P = 2.0 × 10−3), with triple the effective sample size of the largest of those studies10, demonstrating once again the stability of GWAS findings as sufficient sample size and power to discover new loci are reached22,23. We discovered GWS loci in four ancestral groups: EUR, AFR, AMR and EAS. In AFR, two independent SNPs were associated on chromosome 5. The first (rs574008891) was within an intron of the gene that encodes methylcrotonyl-CoA carboxylase subunit 2 (MCCC2). The other significant locus (rs573117193) mapped to an intron in the solute carrier family 36 member 2 (SLC36A2) gene. These specific variants are absent in the other ancestries studied. For AMR, the one risk locus was rare (rs9815757, minor allele frequency (MAF) 0.1%) and mapped in an intergenic region downstream of leucine rich repeat containing 3B (LRRC3B). Finally, for EAS, one locus was associated with CanUD: rs78561048, near semaphorin 6D (SEMA6D). Follow-up analysis in larger samples is needed to assess the robustness of findings, particularly in AMR and EAS. Several variants showed concordant direction of effect across all four stratified ancestral groups (Table 1). For instance, rs10986600, significantly associated in EUR on chromosome 9, was nominally significant (P < 0.05) with same effect direction in AFR (0.04) and AMR (0.03) and significant in the multi-ancestry meta-analysis. This intronic variant of the protein phosphatase 6 catalytic subunit (PPP6C) is an eQTL for PPP6C, a gene linked to various cancers, including skin melanoma and lung squamous cell carcinoma. Multi-ancestry meta-analysis revealed an additional five loci not identified in the stratified analyses. Among them, the lead SNP on chromosome 15, rs147144681, which maps to an intron of the cholinergic receptor nicotinic alpha 3 subunit (CHRNA3) gene, is particularly noteworthy; as reported above, variation in CHRNA2 was among the first variants associated with CanUD and was replicated here. This suggests potential convergence involving the cholinergic system broadly and nicotinic receptors, specifically in the underlying etiology of CanUD. While nicotinic receptors are also associated with tobacco smoking-related traits24, the relative pattern of association for those traits is different from the observations for CanUD—for many smoking-related traits, a chromosome 15 nicotinic receptor cluster is associated with orders of magnitude greater support than other variants, including other nicotinic receptors; for CanUD, CHRNA2 is consistently the strongest association, also by orders of magnitude. We conducted conditional analysis for CHRNA2 and found the conditional P value remained robust following conditioning on smoking initiation20 (Pcond = 4.6 × 10−14). This replicates similar analyses performed by Demontis et al.7 and Johnson et al.10, which showed conditioning on smoking did not affect the CanUD association at this variant. Several other loci near cholinergic receptor subunit genes previously identified for smoking are not significant in our analysis of CanUD (CHRNA4, rs13036436, smoking P = 1.1 × 10−29, CanUD P = 0.97; CHRNA5, rs667282, smoking P = 9.9 × 10−25, CanUD P = 0.043). Conversely, the CHRNA3 variant we find associated with CanUD is not significant for smoking (rs147144681, smoking P = 0.0033, CanUD P = 3.3 × 10−8) (ref. 20).
Genetic correlations were calculated for 1,335 traits to identify genetic overlap with CanUD. Some traits with significant rG were tested for causal inference based on a combination of significant genetic correlation and a prior interest in phenotype (physical activity, multi-site chronic pain, Alzheimer’s disease and SCZ). We identified a bidirectional causal relationship between CanUD and SCZ. At the same time, the MR Egger analysis indicated this was not due to horizontal pleiotropy. This supports similar findings reported previously, confirming previous genetic–epidemiologic studies25 and verifying an important public health risk associated with CanUD. To highlight differences between cannabis use and CanUD, we compared the pattern of genetic correlations across 18 traits, which showed striking differences. CanUD was much more closely associated with psychopathology, recapitulating a general pattern seen with other comparisons of SUD and use traits26. For example, while we observed a substantial negative correlation between CanUD and educational attainment, cannabis use was associated with greater educational attainment. POPCORN was used to generate a cross-covariance score to allow for comparison of traits across ancestries using genetic correlations for EUR and AFR groups (Supplementary Fig. 3). We found a striking similarity for cross trait comparisons for both groups, indicating a similar underlying genomic architecture. This finding supports the possibility that some findings uncovered so far for EUR individuals, recruited in vastly greater numbers for genetic study, will provide some degree of generalizability across human populations.
Chronic pain may be a factor driving CanUD in some individuals, with significant unidirectional evidence for a causal effect of chronic pain27 on CanUD in the MR analysis. Cannabis use has been proposed as a treatment for chronic pain, and there are several clinical trials in progress28. This MR observation suggests that there may be merit in cannabis as a treatment for at least some kinds of pain. The small overall beneficial effect observed requires so many individuals to be treated that harmful effects (such as increased CanUD) also become a significant factor29. Our MR results suggesting that chronic pain has a causal influence on CanUD emphasize the need for follow-up investigations that address whether greater consideration should be given to the adverse effects, rather than just the therapeutic effects among individuals receiving cannabis-based medicines. A similar question arises with opioids, which although often prescribed for pain, can also cause great harm30: namely, what level of risk of CanUD is acceptable given cannabis’ potential to improve quality of life and reduce opioid exposure in chronic pain patients? Our results suggest that harms such as dependence and consequences, reflected in underlying genetics of the trait, may need to be weighed against the potential benefits of cannabis treatment for chronic pain. Future studies should consider this novel relationship to pain31 and clinical efficacy trials are underway.
Cigarette smoking substantially increases the risk of many forms of cancer, including lung cancer, through numerous well-studied mechanisms with established literature dating back more than 60 years32. The influence of cannabis on cancer risk is less well understood; it should be anticipated that these combustion products could have harmful pulmonary impacts—indeed, it would be surprising if smoking tobacco, but not smoking cannabis, increased cancer risk. MR yielded evidence for a unidirectional causal effect of CanUD on lung cancer. This result was robust to conditioning on data from the largest available smoking initiation GWAS but not conditioning on cigarettes per day, both traits that also have causal relationships with lung cancer but far more robust genetic instruments to evaluate this relationship. We do not currently have a way to assess genetic variation associated with the route of cannabis administration, but combustion is by far the most common method in the MVP and other cohorts studied. Given the trend toward increased legalization and usage, this apparent causal association needs to be monitored as it may have profound and underappreciated public health consequences. As the causal relationship with CanUD was not robust to conditioning on cigarettes per day, one probable explanation may be that there is horizontal pleiotropy between these traits in their influence on lung cancer.
Four GWS loci overlapped with TWAS prioritization from the EUR meta-analysis, using eQTL integration from samples of adult33 and fetal34 cortical tissue. These were DALRD3 (both fetal and adult), ERCC8 (fetal), RP11-629G13.1 (adult) and PHLPP2 (adult). The DALRD3 protein product, a DALR anticodon binding domain, forms a complex with the product of METTL2B. Nonsense mutations in DALRD3 are associated with developmental delay and early-onset epileptic encephalopathy35. ERCC8 encodes the excision repair 8, CSA ubiquitin complex subunit, which plays a role in DNA repair and is associated with the developmental disorder Cockayne syndrome36, as well as breast, esophageal and other cancers37,38. RP11-629G13.1 is a long noncoding RNA associated with downregulation of NCAM1 gene expression in multiple myeloma patients39. Significant partitioned SNP-based heritability was observed in fetal but not in the adult cortex, with 4.36% of trait SNP-based heritability explained by 0.12% of the total SNPs near fetal frontal cortex eQTLs. Only 1.77% of CanUD SNP-based heritability was explained using 0.13% of the total SNPs near adult cortex eQTLs. Fetal development may play a role in SUD susceptibility40, and substance use can influence fetal development during pregnancy and health outcomes during childhood41. Although exogenous exposure to cannabis may not occur until years or decades after birth, enriched fetal SNP-based heritability in this study argues a possible role for genetic effects on CanUD in the developing brain independent of exposure. SCZ risk is also modulated by risk factors during fetal development42 and genetic43 and environmental effects (including maternal food deprivation in the first trimester of pregnancy44). Temporal convergence of the initiation of genetic risk effects for both SCZ and CanUD, if validated experimentally, would provide insight into the genetic relationship between these disorders and could relate to a mechanism for the bidirectional risk relationship between cannabis use and SCZ.
gSEM was used to contextualize summary statistics from this project with those from other published GWAS studies. Exploratory and confirmatory factor analyses showed that four factors provide the best fit for the 14 correlated traits included in the analysis. Factors fit mostly into categories that relate to functional impairment (factor 1), impulsivity and risk taking (factor 2), psychopathology (factor 3) and substance dependence (factor 4). CanUD fit best (and strongly) in the substance dependence cluster (factor 4). FTND fit into factors for functional impairment and substance dependence. Suicide attempts fit into functional impairment, impulsivity/risk taking and psychopathology. This is consistent with research showing overlapping pathologies within addiction and shared genetic risk factors between them45.
This study has limitations. The use of electronic health records allows for a large sample of CanUD cases but limits the assessment of subdiagnostic cannabis use in controls. Although we accounted for subdiagnostic cannabis users by excluding them from controls when information was available, these are probably underreported. Future studies of individuals with ascertained cannabis use who do not meet criteria for CanUD would provide more insight into the specific genetic liability to dependence. As the traits of interest were gathered from previously published reports or queries of electronic health records (EHRs) for diagnostic codes, we did not have information regarding tetrahydrocannabinol (THC) blood levels or information on the potency of cannabis at each exposure. If these data were available, study of effects on cannabis potency on dependence and comorbidities would be of great interest. We identified a causal relationship between multi-site chronic pain and CanUD. As pain is a complex trait and different type of pain may interact differently with CanUD, our finding for multi-site chronic pain is not sufficient to draw conclusions about the interaction between CanUD and specific kinds of pain or pain syndromes. Our definition of CanUD was based on any report of abuse or dependence either as an inpatient or outpatient. Participants in this study span a period of changing legal status and increasing use of marijuana, a major secular trend. Given the age of the participants (Supplementary Table 16) and expected time from initial exposure to the development of a use disorder, nearly all participants would have been exposed to cannabis before legalization. The TWAS study did not include ascertainment for CanUD in the individuals who donated brain tissue used for analysis. We discovered GWS loci in ancestral groups, but AFR, AMR and EAS sample sizes were small compared to EUR. We did not perform MR or TWAS analyses in non-European samples because available GWAS and eQTL datasets are still limited in non-European ancestry populations, and cross-ancestry analyses carry risk of biases due to differences in the underlying LD structure between ancestries. More studies are needed of individuals of diverse ancestries to replicate these findings, estimate their robustness and ensure that the benefits provided by these studies are available to all people.
This is the largest genetic study of CanUD so far, including data from multiple international cohorts in more than one million participants and comprising four ancestral groups. We replicate two prior GWS findings while identifying 25 novel loci, and we leverage these novel data to investigate genetic overlap with other traits. We identify a clear difference between cannabis use and CanUD, with genetic liability to CanUD being much more closely associated with psychopathology and disability. We found greater heritability enrichment in fetal than adult brain tissue, supporting an important role of development in laying the biological basis for CanUD. We used MR to assess causal relationships and found evidence of bidirectional causal effects between CanUD and SCZ and unidirectional effects of multi-site chronic pain on CanUD, and of CanUD on lung cancer. Finally, using gSEM, we found that CanUD loads on a latent factor with other substance dependence traits, consistent with clinical observation, genetic epidemiology and prior genetic studies of other SUD traits. In particular, we highlight the possible relationship revealed herein between CanUD and lung cancer risk. This study yields new insights into the genetic architecture of CanUD and how this risk interacts with traits crucial to public health and raises important concerns regarding the potential adverse consequences of the secular trend toward increased cannabis use consequent to legalization.
