|
|
Post by Admin on Jan 8, 2019 18:55:05 GMT
A British geneticist is worried that He Jiankui—the Chinese scientist responsible for the birth of genetically modified human twins—could face the death penalty for corruption and bribery charges. Back in November 2018, Lovell-Badge organized the Second International Summit on Human Genome Editing at the University of Hong Kong. Upon hearing rumors of He’s controversial work, Lovell-Badge decided to invite the 34-year-old scientist and biotech entrepreneur to the summit in hopes of tempering his enthusiasm, or as he told the Telegraph, to “control his urges.” During the summit, He admitted to using the CRISPR/Cas9 gene-editing tool to modify human embryos that are now twin babies.  When He Jiankui went missing back in early December, we suspected big trouble ahead for the rogue scientist, but as Sarah Knapton reports in the Telegraph, his predicament is even worse than we thought. The embattled scientist is reportedly living under armed guard at a state-owned apartment in Shenzhen, China, according to geneticist Robin Lovell-Badge of the Francis Crick Institute in London, who now worries that He could face the death penalty for his indiscretions.  CRISPR-Cas9 was adapted from a naturally occurring genome editing system in bacteria. The bacteria capture snippets of DNA from invading viruses and use them to create DNA segments known as CRISPR arrays. The CRISPR arrays allow the bacteria to "remember" the viruses (or closely related ones). If the viruses attack again, the bacteria produce RNA segments from the CRISPR arrays to target the viruses' DNA. The bacteria then use Cas9 or a similar enzyme to cut the DNA apart, which disables the virus.  The CRISPR-Cas9 system works similarly in the lab. Researchers create a small piece of RNA with a short"guide" sequence that attaches (binds) to a specific target sequence of DNA in a genome. The RNA also binds to the Cas9 enzyme. As in bacteria, the modified RNA is used to recognize the DNA sequence, and the Cas9 enzyme cuts the DNA at the targeted location. Although Cas9 is the enzyme that is used most often, other enzymes (for example Cpf1) can also be used. Once the DNA is cut, researchers use the cell's own DNA repair machinery to add or delete pieces of genetic material, or to make changes to the DNA by replacing an existing segment with a customized DNA sequence. 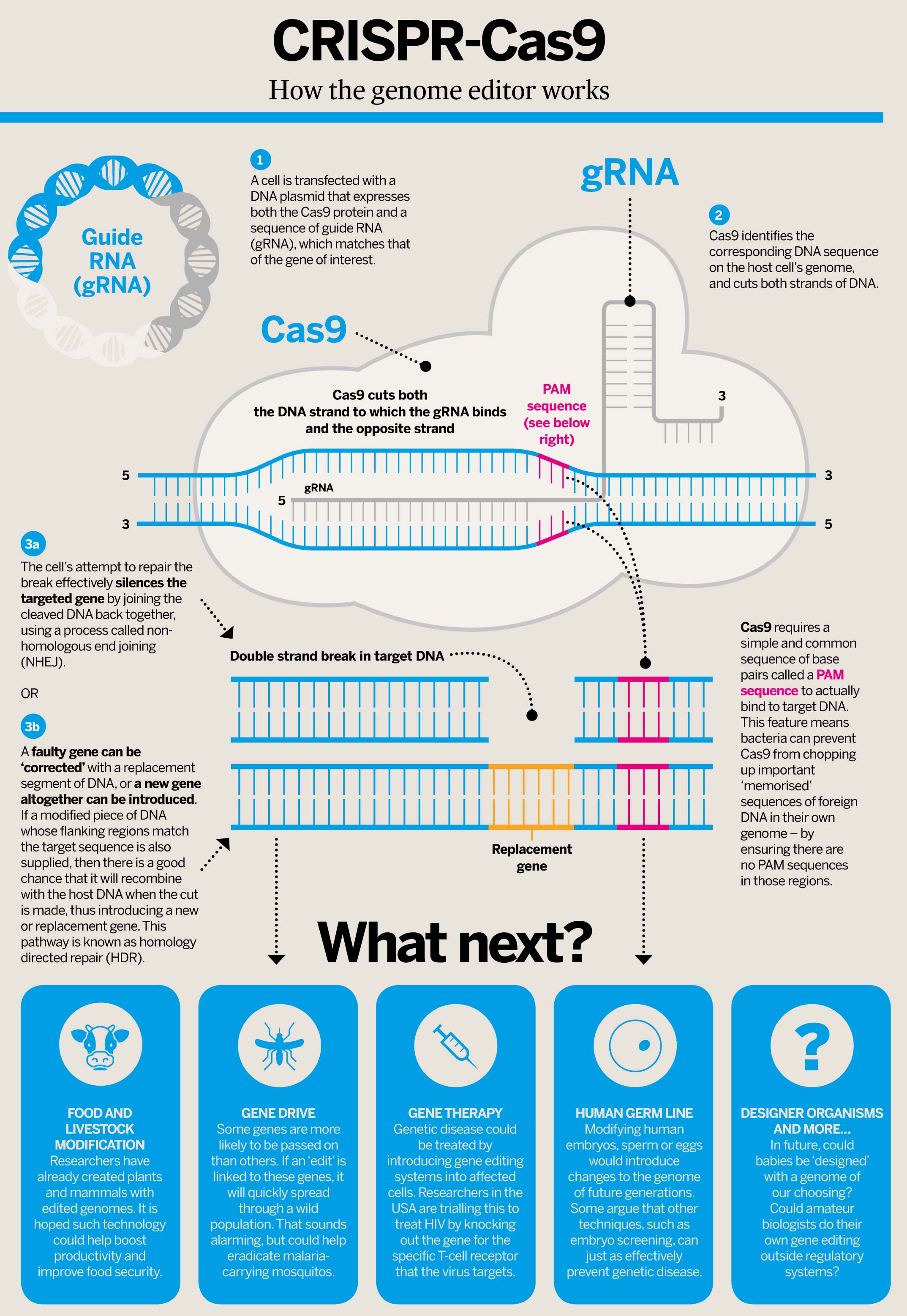 Genome editing is of great interest in the prevention and treatment of human diseases. Currently, most research on genome editing is done to understand diseases using cells and animal models. Scientists are still working to determine whether this approach is safe and effective for use in people. It is being explored in research on a wide variety of diseases, including single-gene disorders such as cystic fibrosis, hemophilia, and sickle cell disease. It also holds promise for the treatment and prevention of more complex diseases, such as cancer, heart disease, mental illness, and human immunodeficiency virus (HIV) infection. Ethical concerns arise when genome editing, using technologies such as CRISPR-Cas9, is used to alter human genomes. Most of the changes introduced with genome editing are limited to somatic cells, which are cells other than egg and sperm cells. These changes affect only certain tissues and are not passed from one generation to the next. However, changes made to genes in egg or sperm cells (germline cells) or in the genes of an embryo could be passed to future generations. Germline cell and embryo genome editing bring up a number of ethical challenges, including whether it would be permissible to use this technology to enhance normal human traits (such as height or intelligence). Based on concerns about ethics and safety, germline cell and embryo genome editing are currently illegal in many countries. |
|
|
|
Post by Admin on Jan 14, 2019 19:30:29 GMT
The world faces a potential antibiotic crisis. A lack of new antibiotic drugs combined with increasing amounts of antibiotic resistant bacteria is setting the stage for a potential pandemic. The calamitous potential has researchers trying all kinds of ways to develop new drugs. From super enzymes to synthetic microbes… heck, even AI has been thrown into the ring.
Now, researchers at the University of Washington-Madison and at the University of California, San Francisco are taking a new approach by repurposing the gene-editing tool CRISPR to study which genes are targeted by particular antibiotics. By observing how antibiotics interact with genes, the researchers hope to find clues to improve existing medicines or develop new ones.
CRISPR is a tool used to edit DNA by slicing out one segment of genes and inserting a new one while the cell is repairing. The new system, developed by UW-Madison professor Jason Peters uses a somewhat modified technique. “Most people, when they think about CRISPR, think about gene editing,” said Peters.
But Peters and team employed a modified version of the technique known as Mobile-CRISPRi. A sort of defanged form of CRISPR, CRISPRi is engineered not to be able to slice DNA but instead sits on top of it blocking off proteins that would otherwise activate certain genes. By selectively blocking different genes the researchers are able to reduce the amount of proteins produced from those genes. They found that if they decreased the amount of protein targeted by an antibiotic, bacteria became much more sensitive to lower levels of the drug. Other researchers could replicate this in order to gather data on thousands of genes at a time.
The Mobile-CRISPRi system is very versatile and easy to use. By exploiting a form of bacterial sex called conjugation, CRISPRi can be easily introduced to different types of bacteria. Peters and team demonstrated this by applying CRISPRi to lab-grown E. Coli bacteria and also a much less commonly encountered kind of bacteria called V. casei which they collected from a rind of French cheese. In both cases the CRISPRi easily adhered to the bacteria’s genetic information.
|
|
|
|
Post by Admin on Jan 15, 2019 17:50:48 GMT
George Church describes going beyond cutting DNA to precise editing. 1. Phage Integrases (Protein and DNA) 2. TAL Deaminases (custom Protein) 3. Chemical Targeting (custom polymer)  For DNA-based target recognition systems, the two major approaches, multiplex automated genome engineering (MAGE) using λ-red recombination and conjugative assembly genome engineering (CAGE) have been widely used in prokaryotes and demonstrated a high degree of multiplexability. Briefly, the λ-red system, similar to recET, originates from phage and when imported into bacterial cells, can stimulate high levels of recombination in the presence of homologous DNA. Although the machinery used in these approaches has not been fully optimized yet for eukaryotic cells, some conjugal transfer has been demonstrated and use of an optimized version of Redβ in human cells has yielded limited success. Moreover, these technologies can be used to edit and propagate large pieces of DNA in bacteria for eventual use as donor DNA molecules for HR in human cells.  |
|
|
|
Post by Admin on Jan 17, 2019 18:13:43 GMT
Gene editing techniques generally involve proteins that cut DNA, such as those employed in CRISPR-Cas9, transcription activator-like effector nucleases (TALENs) and zinc-finger nucleases. The most commonly used Cas enzyme, Cas9, comes from Streptococcus pyogenes—the one that gives you strep throat and was proven viable in mouse and human cells in 2013. The basic process is that the CRISPR molecule is programmed to search for a specific nucleotide sequence amongst the 3 billion in the human genome. Once the correct sequence is identified, CRISPR unwinds the coils of DNA coils and “snips” the sequence out of the strand. DNA strands are then repaired in the case of a gene deletion, or, for an insertion, a new sequence can be included to alter the genome. Performed in an embryonic germ line cell, an egg or a sperm cell, gene “edits” will be part of the genetic code that goes to the next generation. But there can be errors—in other words editing more than intended—with targeting associated with the guide RNA used to target the deletions. It is the presence of these “off-target repeats” that indicates extreme caution and a need for better regulation before techniques like CRISPR can have safe clinical application. As such, we as scientists and society must also balance the potential good associated with new techniques and the prospect of doing something just because we could. Gene editing places great power over altering the fundamental principles of biology, and our whole society needs to part of the discussion on what is okay to do and what is not. And we need to move quickly but not in a hurry. It’s critical to think about the path ahead—which one to take and to where—before we arrive. Scientists and engineers at work right now are working to enable the realization of our common futures. But guiding the implementation of that future is the right and responsibility of us all and cannot be entrusted exclusively to those at work in the field and laboratories, nor to those who attempt to regulate their work, our lawmakers and bureaucrats.  The future we invent can be bright—but there are strings attached. The most important string is that we need input from as many sectors in our society as possible. The decisions that are made will literally affect the future of our species and cannot be made in isolation from our society as a whole. Science works as a machine of chance effects with experimental outcomes; tested against a backdrop of random occurrences and biological evolution is the emergence of chance survival characteristics expanding over millions of years. There is a pace and timing to adaptations. Yet, any modifying of the human germ line—editing sperm or egg cells—has direct implications for the next generation and must be done carefully in light of regulations specifically addressing this kind of experimentation. In many countries there is a de facto moratorium on human germ line and embryo editing because such work is illegal. It is also completely unethical, not least of all because of lack of consent. |
|
|
|
Post by Admin on Jan 23, 2019 18:48:01 GMT
A university in China has severed ties with the scientist who rattled researchers last year after he claimed to have engineered the world’s first gene-edited babies.
The Southern University of Science and Technology (SUSTC) in Shenzhen ended He Jiankui’s employment after government investigators concluded that the scientist’s experiment violated regulations and may incur criminal repercussions. According to the probe, He violated “ethical principles and scientific integrity.”
He claims to have used CRISP-Case 9, a precise gene-editing technique, to alter a pair of embryos to make them impervious to HIV.
The government probe appeared to corroborate He’s claims, which have not been published or independently verified. The scientist executed his experiment “on his own” and “in the pursuit of personal fame,” according to China’s state-run news agency Xinhua.
Based on the probe’s conclusion, SUSTC terminated He’s employment as well as his teaching post and research. He had previously been on unpaid leave, according to the university, which has denied knowledge of his research.
|
|







