|
|
Post by Admin on Oct 24, 2019 17:56:56 GMT
When Niekus and his colleagues used radiocarbon dating to analyze the tar on the flake, they found it was 50,000 years old, dating back to a time before modern humans arrived, they write today in the Proceedings of the National Academy of Sciences.  The tar, preserved by the cold, oxygen-free conditions in sediments several meters beneath the sea floor, might have been an essential element of Stone Age tool kits, says co-author Geeske Langejans, an archaeologist at the Delft University of Technology in the Netherlands. She and her colleagues tried to recreate tool’s manufacture, collecting strips of birch bark, mounding clay over them, and building a fire on top to heat the bark inside to 300°C–400°C for hours. The procedure was hot enough to produce thick tar, as the resinous bark disintegrated. By comparing the chemical composition of the modern tar and its impurities to the ancient tar, Langejans and her team found that the Neanderthals likely used the same procedure.  But making enough tar to adorn even an unremarkable tool was undoubtedly difficult without pottery to collect the hot, pooling tar. “It’s an ugly little piece, not even retouched or shaped,” Langejans says. “That they hafted such a simple flake suggests they used adhesives on a regular basis.” Other evidence suggests Neanderthals used pine resin and bitumen as adhesives to stick stone points to wooden spears. This find and two tarred tools from Italy and Germany suggest our extinct cousins used birch bark tar as well. The discovery also adds to previous work showing Neanderthals could engage in complex tasks, including creating finely crafted stone blades and multipart spears.  Paola Villa, an archaeologist at University of Colorado in Boulder who was not involved in the study, says it is “very good work” that shows the creators of the tools were capable of complex behavior. But, she adds, a literal handful of tools from just three sites is too few to conclude that Neanderthals used birch bark tar routinely. 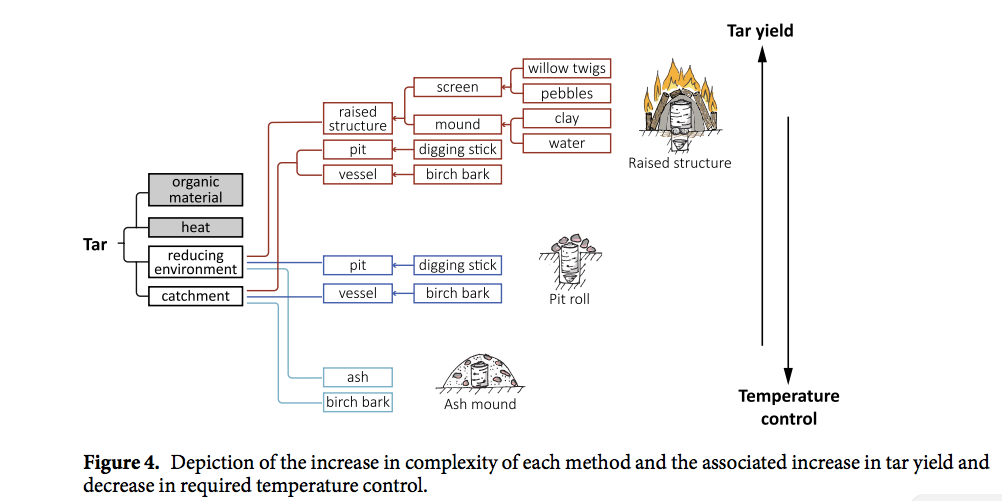 Niekus hopes more finds dragged up from the bottom of the North Sea could change that: “This is the tip of the iceberg,” he says. “Beneath the sea, there’s a lot of sites, and thanks to beach replenishment we can study them.” |
|
|
|
Post by Admin on Nov 8, 2019 18:26:44 GMT
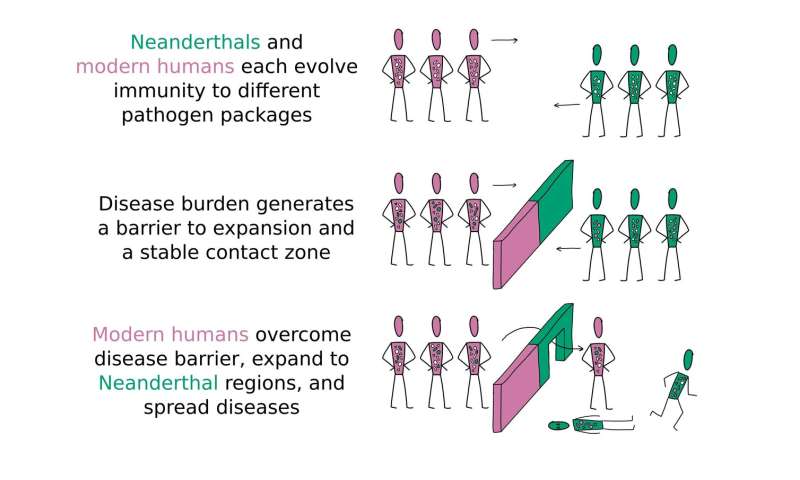 In a new study published in the journal Nature Communications, Greenbaum and his colleagues propose that complex disease transmission patterns can explain not only how modern humans were able to wipe out Neanderthals in Europe and Asia in just a few thousand years but also, perhaps more puzzling, why the end didn't come sooner. "Our research suggests that diseases may have played a more important role in the extinction of the Neanderthals than previously thought. They may even be the main reason why modern humans are now the only human group left on the planet," said Greenbaum, who is the first author of the study and a postdoctoral researcher in Stanford's Department of Biology.  The slow kill Archeological evidence suggests that the initial encounter between Eurasian Neanderthals and an upstart new human species that recently strayed out of Africa—our ancestors—occurred more than 130,000 years ago in the Eastern Mediterranean in a region known as the Levant. Yet tens of thousands of years would pass before Neanderthals began disappearing and modern humans expanded beyond the Levant. Why did it take so long? Employing mathematical models of disease transmission and gene flow, Greenbaum and an international team of collaborators demonstrated how the unique diseases harbored by Neanderthals and modern humans could have created an invisible disease barrier that discouraged forays into enemy territory. Within this narrow contact zone, which was centered in the Levant where first contact took place, Neanderthals and modern humans coexisted in an uneasy equilibrium that lasted tens of millennia. Ironically, what may have broken the stalemate and ultimately allowed our ancestors to supplant Neanderthals was the coming together of our two species through interbreeding. The hybrid humans born of these unions may have carried immune-related genes from both species, which would have slowly spread through modern human and Neanderthal populations. As these protective genes spread, the disease burden or consequences of infection within the two groups gradually lifted. Eventually, a tipping point was reached when modern humans acquired enough immunity that they could venture beyond the Levant and deeper into Neanderthal territory with few health consequences. At this point, other advantages that modern humans may have had over Neanderthals—such as deadlier weapons or more sophisticated social structures—could have taken on greater importance. "Once a certain threshold is crossed, disease burden no longer plays a role, and other factors can kick in," Greenbaum said.  Why us? To understand why modern humans replaced Neanderthals and not the other way around, the researchers modeled what would happen if the suite of tropical diseases our ancestors harbored were deadlier or more numerous than those carried by Neanderthals. "The hypothesis is that the disease burden of the tropics was larger than the disease burden in temperate regions. An asymmetry of disease burden in the contact zone might have favored modern humans, who arrived there from the tropics," said study co-author Noah Rosenberg, the Stanford Professor of Population Genetics and Society in the School of Humanities and Sciences. According to the models, even small differences in disease burden between the two groups at the outset would grow over time, eventually giving our ancestors the edge. "It could be that by the time modern humans were almost entirely released from the added burden of Neanderthal diseases, Neanderthals were still very much vulnerable to modern human diseases," Greenbaum said. "Moreover, as modern humans expanded deeper into Eurasia, they would have encountered Neanderthal populations that did not receive any protective immune genes via hybridization." The researchers note that the scenario they are proposing is similar to what happened when Europeans arrived in the Americas in the 15th and 16th centuries and decimated indigenous populations with their more potent diseases. If this new theory about the Neanderthals' demise is correct, then supporting evidence might be found in the archeological record. "We predict, for example, that Neanderthal and modern human population densities in the Levant during the time period when they coexisted will be lower relative to what they were before and relative to other regions," Greenbaum said. |
|
|
|
Post by Admin on Nov 9, 2019 6:27:23 GMT
Introduction The lineages leading to modern humans (Homo sapiens; henceforth ‘Moderns’) and Neanderthals (H. neanderthalensis) diverged 500–800 kya, with Neanderthals inhabiting Eurasia and Moderns inhabiting Africa1,2,3. Migrating out of Africa, Moderns reached the Levant tens of thousands of years prior to their further spread throughout Eurasia, whereas Neanderthals seem never to have spread south of the Levant4,5,6. The two species most likely interacted in this region, at least intermittently, for extended periods of time, along a fairly narrow front4,7,8,9. This contact is generally believed to have been accompanied by a low level of repeated interbreeding8,9,10,11,12. Later, around 45–50 kya, Moderns spread further into Eurasia, replacing the Neanderthals within a few thousand years, by about 39 kya5,7,13,14 (Fig. 1). 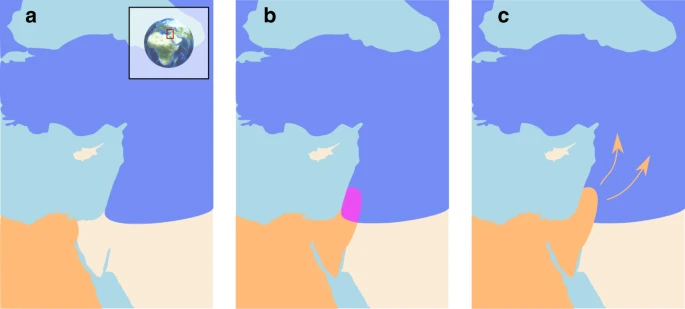 Fig. 1 The replacement of Neanderthals by Moderns has been extensively studied and debated (e.g. refs. 15,16,17,18). Less attention, however, has been given to the fact that contact in the Levant was made much earlier than the initiation of the replacement phase4,7. Therefore, an important question remains open: irrespective of the driving mechanisms of the eventual replacement, what caused the apparent delay in the commencement of the replacement process? This question is especially puzzling because the time frame during which the interaction front was geographically confined to the Levant4,7,19 was much longer than the time frame during which replacement occurred across Eurasia. In this study, we propose that infectious disease dynamics provide a possible solution to this puzzle. We suggest that, following contact, transmissible diseases might have played a prominent role in stabilizing and localizing the inter-species population dynamics in the Levant. We also propose that these dynamics might have resulted in transmissible diseases playing an important role during the later replacement phase. We propose that after several hundred thousand years of largely independent evolution, Neanderthals and Moderns likely acquired immunity and tolerance to different suites of pathogens—a temperate pathogen package in the case of Neanderthals and a tropical pathogen package in Moderns. The re-establishment of contact in the Levant would have resulted in exposure of each species to novel pathogens carried by the other species. In turn, these pathogens could have spread to the new susceptible hosts, placing a considerable disease burden on both species.  Whether the two groups constitute two species or two sub-species is a matter of debate20; for our purposes, all that matters is that they were geographically distinct for a long period of time, and we refer to them as species for convenience. Nevertheless, many genomic studies of Neanderthals and Moderns have detected a signature of introgression (gene flow) between the species, to the extent that Neanderthal sequences may represent 1–3% of present-day non-African modern human genomes11,21,22. That inter-species contact was potentially sufficient to allow for gene flow suggests that disease transmission between the species was likely. Although many of the pathogens that may have been transmitted may not exist today, several genomic studies record potential signatures of events in which pathogens were transmitted between Moderns and Neanderthals, or between Moderns and other archaic humans23,24,25,26,27. Moreover, studies have identified signatures of positive selection on putatively introgressed Neanderthal genes in Moderns, particularly in genomic regions such as the MHC complex that are associated with the immune system2,22,26,28,29,30,31,32. These findings suggest that disease burden due to inter-species pathogen transmission was significant. We use mathematical models of inter-species interaction to explore the possible ecological and demographic consequences of exposure to novel pathogen packages upon inter-species contact, and the coupling of this exposure with immune-related adaptive introgression. Due to the disease burden imposed by contact with the other species, bands of individuals in each species would have been strongly disadvantaged when migrating into regions dominated by the other one, because such migrations would have resulted in increased exposure to novel pathogens. Additionally, disease burden—realized as recurring epidemics, greater endemic pathogen load, or both—would have decreased population densities of both species near the contact front, further reducing the likelihood and motivation for bands of one species to migrate into the range of the other species. The interaction between the two species would thus have been limited to a circumscribed region, which would have been geographically localized by the disease dynamics (Fig. 1). This front may have been constant4,33 or intermittent7,34,35, alternately strenghening and weakening in time and space during the period of contact—in the Levant and perhaps also in the Arabian peninsula36. Some contact between the two species would have continued along this front, resulting in pathogen spillovers between the species, but also in occasional interbreeding through which transmission of immune-related genes would have occurred. Under these circumstances, bands of each species close to the interaction front would gradually have been able to adapt and acquire tolerance to the novel pathogens, through adaptive introgression as well as by selection on de novo immune-related mutations. Eventually, this process would have reduced disease burden, diminishing the effect of disease transmission dynamics and allowing other processes to drive population dynamics. At this point, the barrier to full inter-species contact and cross-regional migration would have been removed, destabilizing the front of interaction and enabling the species dynamics that eventually led to Neanderthal replacement. Disease dynamics, as we will show, are sufficient to explain the extended existence of a stationary interaction front in the Levant, although our analysis does not preclude the importance of additional processes, such as adaptation of the two species to their respective local environments and inter-species competition (e.g. refs. 37,38,39; see the “Discussion” section).  Once the interaction front was destabilized, presumably around 45–50 kya, other processes, previously overshadowed by disease burden, would then have been responsible for the replacement of Neanderthals by Moderns17,18,40, although it has been suggested that disease transmission dynamics could also have played a prominent role in the replacement process31,41,42. Our model supports this possibility. Conditions relating to disease dynamics need not have been symmetric between Moderns and Neanderthals—for example, the Moderns’ tropical pathogen package may have been more burdensome to the Neanderthals than the Neanderthals’ temperate pathogen package was to Moderns, following the pattern of decreasing pathogen burden with latitude43,44. Hence, Moderns may have overcome the disease burden from contact sooner than Neanderthals. This asymmetry would have eventually allowed bands of Moderns to migrate into the Neanderthal regions unhindered by novel transmissible diseases, while carrying contagious diseases to which the Neanderthals were not yet immune. Moreover, after the historical front of interaction was crossed and migration reached deeper into Eurasia, this relative Modern advantage would have increased further, as Neanderthal bands encountered far from the initial contact zone would have been intolerant to the entirety of the novel pathogen package spread by the Moderns. We thus suggest, following patterns that occurred multiple times in the colonial era when two long-separated populations renewed contact27,45,46,47,48, that the replacement of Neanderthals by Moderns may have been facilitated by pathogens to which Moderns were largely immune but to which the Neanderthals were vulnerable. |
|
|
|
Post by Admin on Nov 9, 2019 18:22:44 GMT
Results and discussion Modeling disease transmission and introgression dynamics Our main proposition is that a persistent Modern–Neanderthal front of interaction in the Levant can be explained by disease burden that prevented each species from expanding into the region dominated by the other. We propose that this front could eventually have collapsed due to immune-related adaptive introgression. In order to (i) demonstrate the feasibility of this scenario, (ii) understand the consequences of variation in the details of this process, (iii) investigate the impact of feedback between disease and gene transmission, and (iv) explore the robustness of our proposition, we model disease transmission and introgression dynamics between two species using dynamical-systems models. We first explore a model that bridges two independently treated time-scales, ecological and evolutionary (‘two-time-scales model’), as summarized in Fig. 2. At the ecological time scale, disease spillovers of novel pathogens, whose impact is measured as the proportion of the non-tolerant population that is infected by each epidemic, are modeled in terms of between-species and within-species contact rates (denoted βij, the contact rates from species i to species j), using a well-mixed SIR-modeling framework (Eqs. (3)–(8)). On the evolutionary time scale, the two species respond to disease burden (Di for species i), which we measure as proportional both to the number (or diversity) of novel pathogens to which the species is exposed (Pi) and to the impact of a pathogen at the ecological time scale (Fi); Eqs. (1) and (2). At each time step in the evolutionary time scale, this response is modeled as an adjustment of the contact rates (βij), in proportion to the disease-burden experienced (Eqs. (10)–(13)); for example, in response to the impact of disease that they experience, groups may adjust their tendency to accept individuals from other groups from the same or from different species49. Additionally, also at the evolutionary time-scale, through inter-species contact, immune-related genes are exchanged, decreasing the potential impact of transmissible diseases over time. We assume that the rate of this adaptive introgression is proportional to the inter-species contact rates, and over time reduces the number of novel pathogens Pi to which each species is vulnerable (Eqs. (14) and (15)). 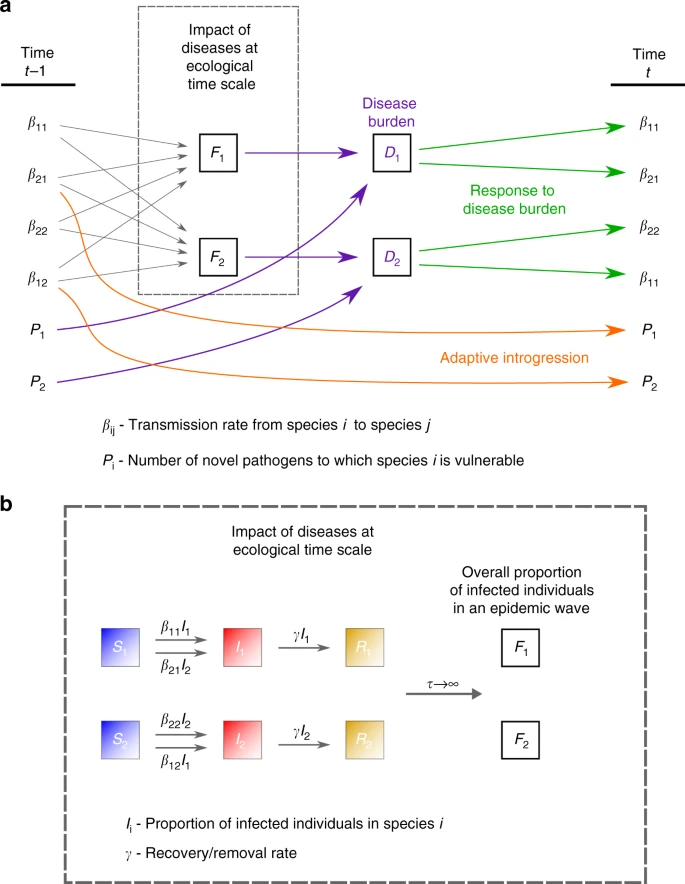 Fig. 2 Symmetric conditions at the time of initial contact We first characterize the general behavior of our model by exploring a scenario in which initial conditions are symmetric for the two species. An outcome of this scenario is presented in Fig. 3. A more thorough exploration of the parameter space, with different parameterizations of the model, appears in Supplementary Note 1, with qualitatively similar results. 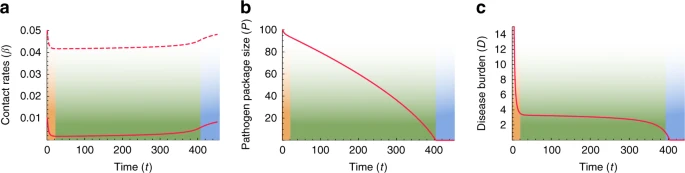 Fig. 3 When the two species first come into contact, high contact rates and vulnerability to many novel pathogens result in high disease burden (Eqs. (1) and (2)), which elicits a rapid response of large effect, and the species lower both within-species and between-species contact rates (Fig. 3, orange phase). Following this initial response, the species maintain stable but low contact rates for an extended period (Fig. 3a, green phase). During this period, the pathogen package Pi is reduced by adaptive introgression (Fig. 3b, green phase; Eqs. (14) and (15)), but disease burden is kept low and close to constant by the continuous minor adjustment of contact rates (βij) made by the species (Fig. 3c, green phase; Eqs. (10)–(13)). We interpret the disease burden at this phase of the dynamics as limiting each species from expanding into regions dominated by the other, thereby leading to geographic localization of the interaction front. This localization is further reinforced by the tendency of bands to isolate themselves, as demonstrated by low contact rates (Fig. 3a, green phase). Eventually, disease burden is removed due to introgression (Di(t)→0 in Eqs. (1) and (2), because Pi(t)→0 in Eqs. (14) and (15)). Once disease burden is removed, the dynamics destabilize, and the species, released from disease burden, begin to recover and return to the initial state that existed before contact was made (Fig. 3, blue phase). This destabilization would then allow other dynamics, previously overshadowed by disease burden, to play out. |
|
|
|
Post by Admin on Nov 10, 2019 18:38:27 GMT
Asymmetric conditions at the time of initial contact It is unlikely that conditions were symmetric at the time that Moderns and Neanderthals came into contact in the Levant, for example with respect to different pathogen co-evolution trajectories in Africa and in Eurasia. We therefore model asymmetric initial conditions, focusing for tractability on one possible aspect of asymmetry at a time. Fig. 4 explores the effect of different initial pathogen package sizes (P2(0) > P1(0)), and asymmetry in initial contact rates is explored in Supplementary Note 1. 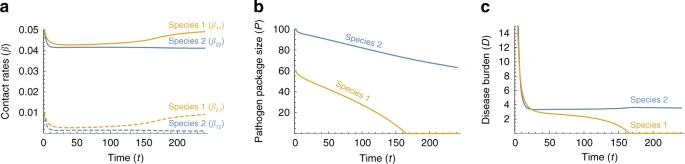 Fig. 4 These dynamics, which are qualitatively similar to Figure 4 with other sources of asymmetries (Supplementary Note 1), mean that the species that was initially less vulnerable, species 1, overcomes disease burden sooner, and is therefore released sooner from the disease limitation enabling expansion into regions dominated by species 2. At the time that species 1 is released from the novel disease burden, species 2 is still vulnerable to many novel pathogens carried by species 1 (Fig. 4b). Feedback between disease transmission and introgression Disease spread between species, in general, depends on the within-species and between-species contact rates, with higher contact rates resulting in greater disease impact. The level of contact between species, and specifically the amount of interbreeding, also determines the expected rate of gene flow, and higher contact rates are expected to result in more rapid adaptive introgression. That both of these phenomena—disease transmission and adaptive introgression—depend on inter-species contact rates, but have opposite effects on the contact rates, generates feedback that complicates prediction of disease and introgression dynamics (Fig. 2). In our model, high disease burden for a species negatively influences contact rates (−aijDj in Eqs. (10)–(13)), and lower between-species contact rates in turn negatively influence the rate of adaptive introgression for that species (−cβij in Eqs. (14) and (15)). Therefore, the model generates a positive feedback that amplifies any initial differences in disease burden over time. In the case of asymmetric pathogen package sizes, the effect of differences in disease burden on the rate of adaptive introgression can be seen in the differing slopes of the pathogen package size trajectories of species 1 and 2 (Fig. 4b). Species 1, which at the time of contact experiences a lower disease burden, also adapts faster to the novel pathogens due to its maintenance of higher incoming between-species contact rates (β21 > β12; Fig. 4a). In the scenario shown, this asymmetry results in the initial difference of P2(0)−P1(0)=40 pathogens in the pathogen package sizes at the time of contact (the units could be different, such as pathogen diversity, but we focus on the relative outcomes without attention to the interpretation of the units), growing to about P2(t)−P1(t)=75 pathogens by the time species 1 has overcome disease burden (Fig. 4b). Another implication of this feedback is observed when species 1 is released from disease burden, i.e. when P1=0 is reached (Fig. 4b). At this point, species 1 begins to increase its contact rates (Fig. 4a), and in so doing increases the impact of disease in the two-species system (Eqs. (3)–(9)). However, at this point, only species 2 is vulnerable to the novel disease, and therefore, only species 2 is forced to respond by further reducing contact rates (Fig. 4c), further slowing the rate of adaptive introgression into species 2. The consequence of the feedback is, therefore, that the species that overcomes disease burden sooner exerts an additional disease pressure on the other species during the destabilization phase. Alternative model The model presented above focuses on behavioral responses to disease burden. However, given that the model requires a seemingly complex behavior—a population must consciously or unconsciously modulate its interaction with other populations based on the amount of disease burden it experiences—it is worthwhile considering whether alternative models that emphasize other factors can produce similar dynamics. We explored an alternative model, which focuses on demographic processes and endemic diseases. In this ‘single-time-scale model’, disease dynamics, adaptive introgression, and population dynamics occur in parallel, over a single time scale, as described by Eqs. (16)–(22). Here, we assume that the entire population of each species is infected by a suite of endemic pathogens, to which it has evolved tolerance. The pathogens inflict no harm on the hosts in the source species, but they are novel to the other species, and therefore cause increased mortality αi(t) among infected individuals in the non-source species, who number Ii(t). In this model, population densities, Ni(t), are determined by density-dependent growth rates and by mortality rates (Eqs. (17) and (18)). We assume that between-species and within-species encounters are determined according to a process similar to Brownian motion of colliding particles, and therefore, contact intensities are modeled as proportional to the population densities (Eqs. (19) and (20)). In this model, response to disease burden is determined purely through demographic effects. Adaptive introgression has the effect of decreasing the mortality rates associated with the novel pathogens, and the introgression rates, like the disease transmission rates, are proportional to the inter-species contact intensity (Eqs. (21) and (22)). Under symmetric initial conditions, the dynamics are similar to those observed in the two-time-scales model: an initial phase of high disease burden resulting in a decrease in population densities (Fig. 5, orange phase), followed by a long phase of low but stable disease burden with low population densities (Fig. 5, green phase), and eventual destabilization and population growth as disease burden is overcome (Fig. 5, blue phase). 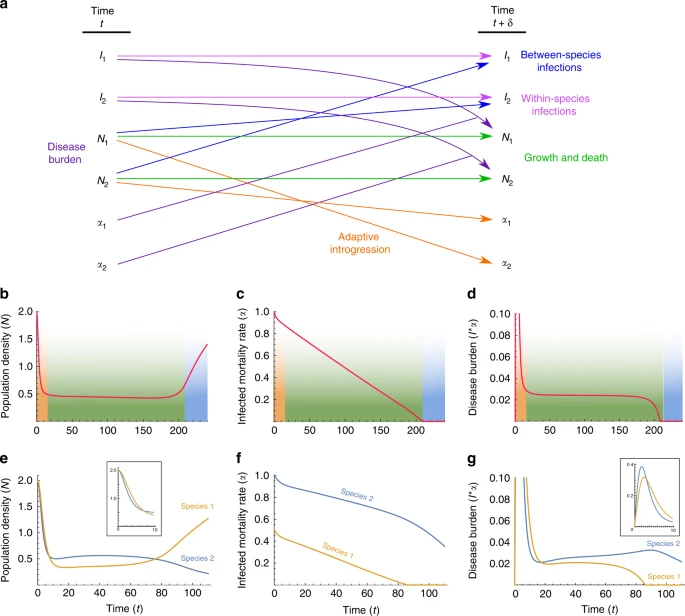 Fig. 5 When the initial conditions are asymmetric with respect to the pathogen packages, α2(0) > α1(0), species 2 suffers higher mortality than species 1 due to novel pathogens. In this case, we observe that the two species initially respond strongly to disease burden and that population densities are reduced (Fig. 5d). A long stable phase of low population densities then follows. As is seen in the two-time-scales model, species 1 is released from disease burden sooner than species 2, when it begins to increase in density towards the initial density at the time of contact (Fig. 5e–g). With initial asymmetry in population densities (N2(0) > N1(0)), but equal pathogen packages (α1(0)=α2(0)), dynamics are qualitatively similar to those with asymmetry in pathogen packages, with the initially less dense species 1 overcoming disease burden sooner in the scenarios examined (Supplementary Note 1). In the two-time-scales model, the separation of the ecological and evolutionary time scales simplifies the dynamics, since the dynamics in the ecological time scale are connected to the evolutionary time scale through the single summarizing parameter of disease impact, Fi(t). In the single-time-scale model, however, interaction between disease, introgression, and demography occurs on the same time scale, resulting in more complex feedback among the different processes. Therefore, whether the consequence of the feedback is increased or decreased asymmetry at the time that species 1 overcomes disease burden depends on the source of the asymmetry and the parametrization of the model. In the scenario in Fig. 5, and with many of the other scenarios we explored (Supplementary Note 1), adaptive introgression in species 1 is more rapid than in species 2, and the differences between the disease-related mortality rates are increased at the time that species 1 overcomes disease burden, relative to the initial asymmetry; however, unlike in the two-time-scales model, this outcome does not always arise, and in some cases, the differences in mortality rates between the species are lower at the time species 1 overcomes disease burden than at the beginning of the dynamics (e.g., Supplementary Figs. 8K and 10K). Once species 1 is released from disease burden and increases in size, the feedback is similar to that in the two-time-scales model, since the increased population densities cause higher disease transmission in the two-species system, which at this point is detrimental only to species 2. |
|


















