|
|
Post by Admin on Apr 5, 2023 18:01:07 GMT
Methods Summary DNA was extracted from the La Braña 1 tooth specimen with a previously published protocol2. Indexed libraries were built from the ancient extract and sequenced on the Illumina HiSeq platform. Reads generated were mapped with BWA27 to the human reference genome (NCBI 37, hg19) after primer trimming. A metagenomic analysis and taxonomic identification was generated with the remaining reads using BLAST 2.2.27+ and MEGAN428 (Extended Data Fig. 9 and Supplementary Information). SNP calling was undertaken using a specific bioinformatic pipeline designed to account for ancient DNA errors. Specifically, the quality of misincorporations likely caused by aDNA damage was rescaled using the mapDamage2.0 software29, and a set of variants with a minimum read depth of 4 was produced with GATK30. Analyses including PCA10, Outgroup f316 and D statistics17 were performed to determine the population affinities of this Mesolithic individual (Supplementary Information). Extended Data Figure 1  Alignment and coverage statistics of the La Braña 1 genome a, Alignment summary of La Braña 1 sequence data to hg19 assembly. b, Coverage statistics per chromosome. The percentage of the chromosome covered by at least one read is shown, as well as the mean read depth of all positions and positions covered by at least one read. c, Percentage of the genome covered at different minimum read depths. Extended Data Figure 2  Damage pattern of La Braña 1 sequenced reads Frequencies of C to T (red) and G to A (blue) misincorporations at the 5′ end (left) and 3′ end (right) are shown for the nuclear DNA (a) and mtDNA (b). Fragment length distribution of reads mapping to the nuclear genome (c) and mtDNA genome (d). Coefficients of determination (R2) for an exponential decline and damage fractions (λ) are provided for the four different datasets. Extended Data Figure 3  Genetic affinities of the La Braña 1 genome a, Principal Component Analysis (PCA) of the La Braña 1 SNP data and the 1000G Project European individuals. b, PCA of La Braña 1 vs Omni world-wide data. Continental terms refer to each Omni population groupings as follows. Africans: Yoruba and Luyha. Asians: Chinese (Beijing, Denver, South, Dai), Japanese and Vietnamese. Europeans: Iberians, Tuscans, British, Finns and CEU; and Indian Gujarati from Texas. c, Each panel shows PC1 and PC2 based on the PCA of one of the ancient samples with the merged POPRES+FINHM sample, prior to Procrustes transformation. The ancient samples include the La Braña 1 sample, four Neolithic samples from Skoglund et al.3 and Keller et al.1. |
|
|
|
Post by Admin on Apr 6, 2023 20:29:39 GMT
Extended Data Figure 4  Allele sharing analysis Each panel shows the allele sharing of a particular Neolithic sample from Skoglund et al.3 and Keller et al.1 with the La Braña 1 sample. The sample IDs are presented in the upper left of each panel (Ajv52, Ajv70, Ire8, Gok4, and Ötzi). In the upper right of each panel, the Pearson’s correlation coefficient is given with associated p-value. Extended Data Figure 5  Pairwise outgroup f3 statistics a, Sardinian vs. Karitiana. b, Sardinian vs. Han. c, La Braña 1 vs. Mal’ta. d, Sardinian vs Mal’ta. e, La Braña 1 vs. Karitiana. The solid line is the y = x line. Extended Data Figure 6  Analysis of heterozygosity a, Heterozygosity distributions of La Braña 1 and modern individuals with similar coverage from the 1,000 Genomes Project (using 1 Mb-windows with 200 Kb overlap). GBR: Great Britain, CEU: Northern-Western European ancestry, IBS: Iberians, FIN: Finns, TSI: Tuscans, YRI: Yorubans, LWK: Luhya, CHB: Han Chinese, JPT: Japanese. b, Heterozygosity values in 1-Mb windows (with 200 kb overlap) across each chromosome. |
|
|
|
Post by Admin on Apr 20, 2023 15:50:17 GMT
Certain stem cells have a unique ability to move between growth compartments in hair follicles, but get stuck as people age and so lose their ability to mature and maintain hair color, a new study shows. Led by researchers from NYU Grossman School of Medicine, the new work focused on cells in the skin of mice and also found in humans called melanocyte stem cells, or McSCs. Hair color is controlled by whether nonfunctional but continually multiplying pools of McSCs within hair follicles get the signal to become mature cells that make the protein pigments responsible for color. Published in the journal Nature on April 19, the new study showed that McSCs are remarkably plastic. This means that during normal hair growth, such cells continually move back and forth on the maturity axis as they transit between compartments of the developing hair follicle. It is inside these compartments where McSCs are exposed to different levels of maturity-influencing protein signals. 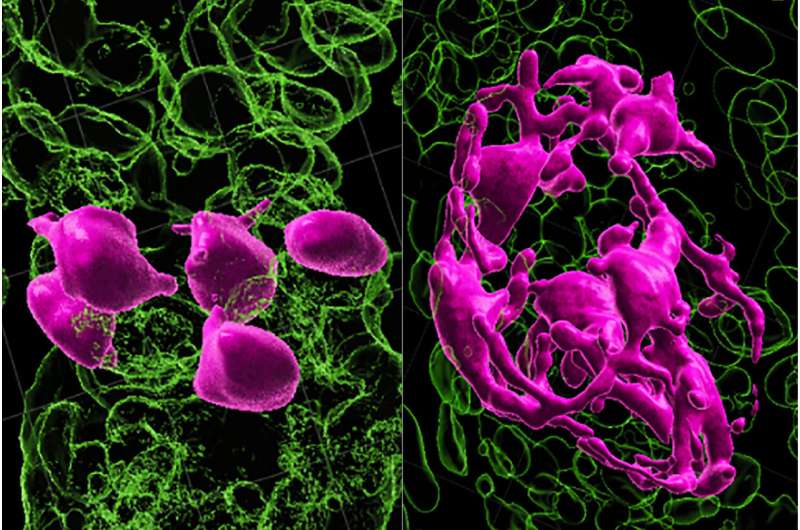 Specifically, the research team found that McSCs transform between their most primitive stem cell state and the next stage of their maturation, the transit-amplifying state, depending on their location. The researchers found that as hair ages, sheds and then repeatedly grows back, increasing numbers of McSCs get stuck in the stem cell compartment called the hair follicle bulge. There, they remain, do not mature into the transit-amplifying state and do not travel back to their original location in the germ compartment, where WNT proteins would have prodded them to regenerate into pigment cells. "Our study adds to our basic understanding of how melanocyte stem cells work to color hair," said study lead investigator Qi Sun, Ph.D., a postdoctoral fellow at NYU Langone Health. "The newfound mechanisms raise the possibility that the same fixed-positioning of melanocyte stem cells may exist in humans. If so, it presents a potential pathway for reversing or preventing the graying of human hair by helping jammed cells to move again between developing hair follicle compartments." Researchers say McSC plasticity is not present in other self-regenerating stem cells, such as those making up the hair follicle itself, which are known to move in only one direction along an established timeline as they mature. For example, transit-amplifying hair follicle cells never revert to their original stem cell state. This helps explain in part why hair can keep growing even while its pigmentation fails, says Sun. Earlier work by the same research team at NYU showed that WNT signaling was needed to stimulate the McSCs to mature and produce pigment. That study had also shown that McSCs were many trillions of times less exposed to WNT signaling in the hair follicle bulge than in the hair germ compartment, which is situated directly below the bulge. In the latest experiments in mice whose hair was physically aged by plucking and forced regrowth, the number of hair follicles with McSCs lodged in the follicle bulge increased from 15% before plucking to nearly half after forced aging. These cells remained incapable of regenerating or maturing into pigment-producing melanocytes. The stuck McSCs, the researchers found, ceased their regenerative behavior as they were no longer exposed to much WNT signaling and hence their ability to produce pigment in new hair follicles, which continued to grow. By contrast, other McSCs that continued to move back and forth between the follicle bulge and hair germ retained their ability to regenerate as McSCs, mature into melanocytes, and produce pigment over the entire study period of two years. "It is the loss of chameleon-like function in melanocyte stem cells that may be responsible for graying and loss of hair color," said study senior investigator Mayumi Ito, Ph.D., a professor in the Ronald O. Perelman Department of Dermatology and the Department of Cell Biology at NYU Langone Health. "These findings suggest that melanocyte stem cell motility and reversible differentiation are key to keeping hair healthy and colored," said Ito, who is also a professor in the Department of Cell Biology at NYU Langone. Ito says the team has plans to investigate means of restoring motility of McSCs or of physically moving them back to their germ compartment, where they can produce pigment. For the study, researchers used recent 3D-intravital-imaging and scRNA-seq techniques to track cells in almost real time as they aged and moved within each hair follicle. More information: Mayumi Ito, De-differentiation maintains melanocyte stem cells in a dynamic niche, Nature (2023). DOI: 10.1038/s41586-023-05960-6. www.nature.com/articles/s41586-023-05960-6 |
|
|
|
Post by Admin on Apr 21, 2023 16:49:10 GMT
Dedifferentiation maintains melanocyte stem cells in a dynamic niche Abstract For unknow reasons, the melanocyte stem cell (McSC) system fails earlier than other adult stem cell populations1, which leads to hair greying in most humans and mice2,3. Current dogma states that McSCs are reserved in an undifferentiated state in the hair follicle niche, physically segregated from differentiated progeny that migrate away following cues of regenerative stimuli4,5,6,7,8. Here we show that most McSCs toggle between transit-amplifying and stem cell states for both self-renewal and generation of mature progeny, a mechanism fundamentally distinct from those of other self-renewing systems. Live imaging and single-cell RNA sequencing revealed that McSCs are mobile, translocating between hair follicle stem cell and transit-amplifying compartments where they reversibly enter distinct differentiation states governed by local microenvironmental cues (for example, WNT). Long-term lineage tracing demonstrated that the McSC system is maintained by reverted McSCs rather than by reserved stem cells inherently exempt from reversible changes. During ageing, there is accumulation of stranded McSCs that do not contribute to the regeneration of melanocyte progeny. These results identify a new model whereby dedifferentiation is integral to homeostatic stem cell maintenance and suggest that modulating McSC mobility may represent a new approach for the prevention of hair greying. Main Mammalian tissue regeneration largely depends on the capacity of adult stem cells to differentiate. Stem cell differentiation is generally viewed as unidirectional and follows the hierarchical model originally established through the study of haematopoietic stem cells9,10,11. This theory proposes that stem cells (undifferentiated state) have two distinct fates: one to sustain themselves through self-renewal and the second to produce transit-amplifying (TA) progeny (intermediate differentiated state) that ultimately give rise to functional differentiated cells during tissue regeneration12,13,14. In this model, the life-long durability of self-renewing tissues is typically sustained by a functionally and molecularly heterogeneous pool of stem and progenitor cells. The organization of the McSC system, responsible for hair pigmentation, is thought to parallel that of hair follicle stem cells (HFSCs)5,6,7,8. McSCs are located in the bulge and hair germ (HG) area in telogen-phase hair follicles (HFs)4,5, where they are surrounded by HF epithelial stem cells (bulge cells)14 and progenitor cells (HG cells)15,16 that constitute to the McSC niche. At the onset of the anagen growth phase, McSCs regenerate differentiated melanocytes that migrate downwards into the hair bulb, where they produce pigment for the hair. Similar to HG epithelial cells, HG McSCs activate WNT signalling and undergo differentiation at the onset of regeneration7. Furthermore, McSCs in the bulge cycle more slowly than those in the HG during HF regeneration6. On the basis of these studies, McSCs in the bulge are postulated to represent long-term stem cells6. However, their distinct functions and self-renewal capacities have yet to be characterized. Despite the close relationship between HFSCs and McSCs, there are disparities in their durability over time: McSCs become exhausted earlier than HFSCs in most animals and humans, which results in hair greying during ageing1,2,3. The high prevalence of hair greying suggests that there may be specific disadvantages in the long-term maintenance of McSCs. HG McSCs can regenerate all melanocyte compartments To better understand the hierarchical structure of the McSC system, we quantified the distribution of McSCs before HF regeneration (that is, in the telogen phase). McSCs are defined as DCT+ melanocytes located at the level of the bulge and slightly below the bulge area4. DCT is a melanocyte lineage marker expressed by all melanocytes during the hair cycle4,8,17. Consistent with previous microscopy analyses4,5, DCT+ McSCs were found in the bulge and the HG. However, a new volumetric analysis with 3D imaging of whole telogen HFs from DctLacZ and DctrtTA;tetOH2B-GFP reporter mice17,18 revealed that most HFs lacked any McSCs within the CD34+ bulge. Rather, most McSCs were concentrated in the P-cadherin+ HG (Fig. 1a and Extended Data Fig. 1). Even in the HFs that contained rare bulge McSCs, the majority were located in the HG (Extended Data Fig. 1a). In agreement, labelling of Wnt1cre;RosaLSL-tdTomato mice, in which neural-crest-derived cells, including the melanocyte lineage, are labelled by the fluorescent protein tdTomato showed that most tdTomato+ McSCs were concentrated in the HG7 (Extended Data Fig. 1c). These results suggest that hair pigmentation and McSC maintenance may solely rely on McSCs in the HG. Fig. 1: HG McSCs possess self-renewal ability. 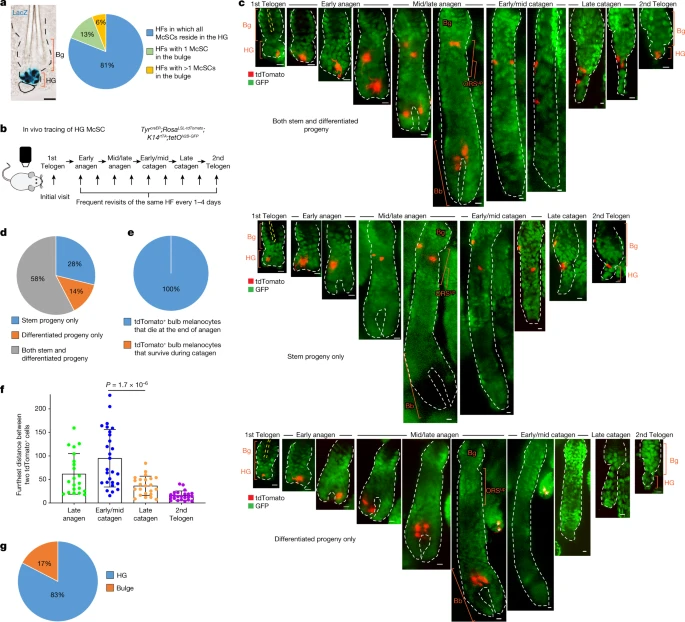 a, Left, bright-field image of a X-gal-stained HF from a DctLacZ mouse at telogen. Right, quantification of McSCs in specified locations in telogen HFs from DctLacZ mice. N = 52 single, whole HFs from 3 mice. b, Timeline of in vivo imaging of single HG McSCs in TyrcreER;RosaLSL-tdTomato;K14rtTA;tetOH2B-GFP mice. HFs containing single HG tdTomato-labelled cells were identified at the first telogen stage and then revisited at indicated stages. c, Live z-stack images of three representative examples of tdTomato+ HG McSCs that undergo distinct fates. K14+ epithelial cells (GFP+) are in green. See Extended Data Fig. 2 for additional examples. Yellow dashed line outlines club hair. White asterisk marks tdTomato+ cell in unrelated HF (bottom). d, Quantification of distinct fates of HG McSCs. N = 59 HFs from 7 mice. e, Percentages of tdTomato+ bulb MCs that die at the end of anagen or survive during the catagen phase. N = 20 HFs from 2 mice. f, Bar graph showing the furthest distance between two tdTomato+ cells in the bulge/ORSup (at late anagen and early/mid catagen) and bulge/HG (at late catagen and second telogen). Data are presented as the mean ± s.d. P values (one-way analysis of variance (ANOVA) with Bonferroni multiple comparison test) are indicated, with 95% confidence interval at 35.81–81.13. N = 21 (late anagen), 27 (early/mid catagen) or 23 (late catagen and telogen) HFs from 2 mice. g, Percentages of tdTomato+ McSCs residing in bulge or HG at second telogen. N = 60 HFs from 6 mice. Dashed black or white lines outline the epithelial–dermal boundary (a,c). Scale bars, 20 μm (a) or 10 μm (c). Bb, bulb; Bg, bulge. |
|
|
|
Post by Admin on Apr 22, 2023 16:50:38 GMT
To verify that HG McSCs have self-renewal ability, we performed fate mapping of telogen HG McSCs using in vivo imaging. We used a mouse model in which McSCs are genetically labelled with tdTomato, and K14+ epithelial cells express GFP, which outlines the HF structure (TyrcreER;RosaLSL-tdTomato;K14rtTA;tetOH2B-GFP mice19,20,21,22) (Fig. 1b). TyrcreER mice are broadly utilized to target melanocytes, including DCT+ McSCs7,8,19,23,24. For clonal analysis, we treated the mice with a low dose of tamoxifen during telogen and identified HFs with only one labelled McSC in the HG compartment. Revisitation of the same HF during anagen (Methods and Extended Data Fig. 2a) showed that HG McSCs labelled during telogen gave rise to mature melanocytes in the hair bulb that die at the end of anagen25 (Fig. 1c–e and Extended Data Fig. 2b). Notably, we also discovered that labelled McSCs migrated to the bulge and upper outer root sheath (bulge/ORSup) stem cell niche during anagen and exhibited self-renewal capacity that enabled their persistence (Fig. 1c,d and Extended Data Fig. 2b). During late catagen, most McSCs in the bulge/ORSup were aggregated in the lower follicle (Fig. 1f, Extended Data Fig. 3). By the next telogen phase, they had primarily homed back to the HG compartment (Fig. 1c,g and Extended Data Fig. 2b ). These results demonstrate that unlike the HFSC system, in which HG epithelial cells disappear after proliferation and differentiation15,26, melanocytes residing within the HG possess self-renewal ability. Trajectory of McSC differentiation at anagen onset Previous studies have shown that WNT proteins in the HG promote differentiation of epithelial cells and melanocytes following induction of HF regeneration7. Epithelial HG cells then function as TA cells that proliferate and differentiate without self-renewal capacity15,26. To investigate how McSCs can retain their stemness while being aggregated in such a pro-differentiation environment, we examined the differentiation status of HG McSCs. In the quiescent telogen stage, McSCs displayed the compact oval or bipolar shape of undifferentiated melanocytes. However, they showed marked changes during early anagen (anagen II), when TA cells emerge in this HG compartment4,5,6, with all melanocytes in the HG developing a dendritic appearance reminiscent of differentiated melanocytes27 (Fig. 2a,b). In more than 90% of the HFs examined, all melanocytes had dendritic morphology during this phase (Fig. 2b). Live imaging showed that the majority of HG McSCs had transformed into a dendritic morphology within the niche before their initial division (Fig. 2c and Extended Data Fig. 4). Daughter cells were also dendritic, which suggested that morphological changes represent an early feature of McSC activation (Fig. 2c and Extended Data Fig. 4). This morphological change was transient, and McSCs retracted dendrites after they migrated from the HG to the bulge/ORSup (Fig. 2c,d). Fig. 2: McSCs in the HG initiate a differentiation programme during early anagen. 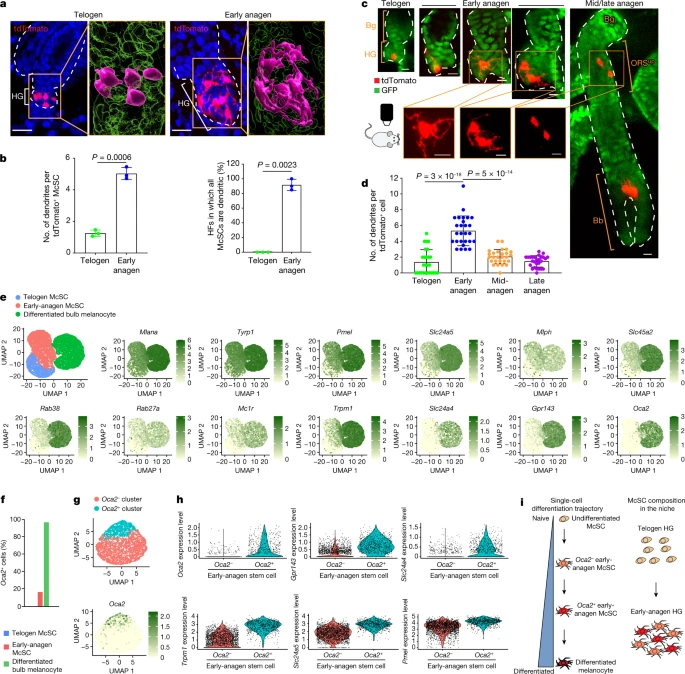 Single-cell RNA sequencing (scRNA-seq) comparisons of telogen and early-anagen McSCs and mature bulb melanocytes purified by fluorescence-activated cell sorting (FACS) verified that genes involved in melanocyte dendrite formation28,29, such as Rac1, were upregulated in early anagen, similar to that observed in mature bulb melanocytes of anagen HFs (Extended Data Fig. 5a–c). Given that the definition of differentiation in biology is “a process by which a less specialized cell matures to fulfil the function of the lineage”30, we examined pigmentation genes as reliable differentiation markers. FeaturePlot analysis showed that numerous pigmentation genes, absent from telogen McSCs, were detected in most early-anagen McSCs (Fig. 2e). Furthermore, a comparison of proliferative and nonproliferative early-anagen McSCs revealed similar signatures, which suggested that differentiation may occur independent of the proliferative state (Extended Data Fig. 5d,e). Nonetheless, a distinct set of genes was expressed by both telogen and early-anagen HG McSCs, but downregulated in bulb melanocytes (Extended Data Fig. 5f,g). Some of these genes have potential stem cell relevance, with functions related to renewal and survival31,32. Pseudotime analysis confirmed that early-anagen McSCs reside between telogen McSCs and bulb melanocytes (Extended Data Fig. 5h). |
|












